Aquarium Redox Balance
Fish Health | Potential & Reduction | rH
Fish Health | Potential & Reduction | rH
Overview
ALL INFORMATION contained herein is copyrighted research and may only be used with permission, in particular to promote products for sale outside of AAP.
PLEASE NOTE: This article is the well-known established authority on the subject of Aquarium Redox and rH (as clearly proven via searches and the history of this subject). All research points to this website as the original and the use of ANY MATERIAL from this article, if even just the subject matter without citing the author here (Carl Strohmeyer) is a violation of the DMCA and will be reported and other legal actions taken if not cited properly. Some reputable websites such as "Aquarium Article Digest" and "Aquarium Wiki" have properly cited this article.
As well, while many will use the fact that the author of this article uses this information to promote products that aid in a healthy aquarium redox, some use this to make Ad Hominem arguments against this information-based products promoted. HOWEVER, if one reads this article in full as well as the history of the author and his extremely in-depth aquarium keeping experience, it is noteworthy that the research came first as to explain contradictory results he was getting in the 1000s of aquariums under his care!
PLEASE NOTE: This article is the well-known established authority on the subject of Aquarium Redox and rH (as clearly proven via searches and the history of this subject). All research points to this website as the original and the use of ANY MATERIAL from this article, if even just the subject matter without citing the author here (Carl Strohmeyer) is a violation of the DMCA and will be reported and other legal actions taken if not cited properly. Some reputable websites such as "Aquarium Article Digest" and "Aquarium Wiki" have properly cited this article.
As well, while many will use the fact that the author of this article uses this information to promote products that aid in a healthy aquarium redox, some use this to make Ad Hominem arguments against this information-based products promoted. HOWEVER, if one reads this article in full as well as the history of the author and his extremely in-depth aquarium keeping experience, it is noteworthy that the research came first as to explain contradictory results he was getting in the 1000s of aquariums under his care!
Although not a well-known process among many aquarists, with much misunderstanding of both sides of the equation by even some advanced aquarium keepers; the implications of Redox for a healthy aquarium are quite far reaching, especially now with peer reviewed research into the correlation of rH, and thus important for any aquarist considering moving from basic aquarium (or pond) keeping to advanced to understand.
Even the average aquarist should consider this water parameter when all other parameters check out, yet fish continue to be susceptible to disease. Not only do my years of research and observations with 1000s of aquariums bears out this is an important subject to consider, but growing research in human disease resistance does as well (Redox is constantly discussed in medical research papers, and the higher Redox level previous thought of as healthy have been shown otherwise).
As research grows, knowing what can affect oxidation and reduction which are both important in their own way can help an aquarium keeper deal better with sick fish or an aquarium that has a sudden build up of organics. Even lighting and quality of light is showing to have an affect on Redox based on tests!
However, some in the aquarium keeping community still seem to be in the dark as per this growing documented research.
Redox, also known as Redox Potential, oxidation potential, & ORP (oxidation reduction potential) describes the ability for the loss of an electron by a molecule, atom or ion to the gain of an electron by another molecule, atom or ion. Without this ability to gain electrons, many minerals cannot be absorbed and properly assimilated, especially in times of stress.
So, it is very important to keep a healthy Redox Balance via both sides of the Redox equation: (a) Normal oxidizers such as proper/optimum dissolved oxygen levels. (b) To counter oxidative stress (often artificially induced in our aquariums); via proper positively charged mineral levels (such as Calcium and Magnesium) and even level 1 or higher UV Sterilization.
Often, we as aquarium keepers perform methods to counter oxidative stress without even knowing it, this includes GH boosters used in the "Estimative Index of Dosing" for planted aquariums. In fact, this is how I got started in this research starting in the 1980s, as my observations between my 1000s of client aquariums were showing different results with different aquarium keeping practices, and often these results took months if not years to observe the often subtle but sometimes substantial difference.
Basically we are attempting to provide enough oxidation to provide ample oxygen, allow for biological organic waste breakdown, and not allow too much bacteria in the water column. WHILE AT THE SAME TIME, not add oxidative stress to inhabitants in the aquarium and allow optimum osmoregulation too.
Many in the past have stated exact numbers are the "best" Redox reading for an aquarium (myself included in the past). My research has found this to be incorrect. In some aquariums, a higher Redox of 350 mV may be a good Redox to obtain (for oxidation) when high organics and decomposition is lowering water quality. HOWEVER, for a new or established aquarium that is properly functioning this is not a number you should be necessarily "shooting" for correct Redox balance, as high Redox does not necessarily equate to a healthy aquarium.
A Better simple explanation of "Healthy Redox" is a "Balanced Redox" that recognizes the need for oxidizers to keep clean well oxygenated aquarium water (especially during treatment) while also recognizing that reduction is NECESSARY to balance out oxidizers such as nitric acid that long term affect fish health. So please read on to better understand!
It is also noteworthy that there is a correlation between Redox Potential and pH (Reference: University of Florida; Biochemistry of Wetlands.
Redox also relates to water changes and the amount/frequency thereof. This article has information about using Potassium Permanganate in a very simple (& fun) test to determine the health of your aquarium before and after a water change: Aquarium Cleaning; Reasons (See Part 7, Redox Section).
We should note that we have both slow and fast Redox reducers and slow and fast Redox oxidizers.
Generally in a healthy aquarium regular use of "fast" oxidizers or reducers is not necessary (which the exception of occasional water conditioner use for chlorine reduction). Fast reducers or oxidizers also generally are relatively temporary, although chloramines are definitely a fast reducer that is not temporary.
Examples include:
Slow Reducers: AAP Wonder Shells, use of constant slow flow through water change with mineralized water, some plant ferts, oolitic sand in reaction chamber.
Fast Reducers: Most water conditioners, including Vitamin C or powdered AAP Wonder Shells
Slow (or low level) to Medium Oxidizers: Products such as Methylene Blue (at high concentrations... can be a reducer at low therapeutic levels), Melafix, Herbtana, Artemiss, activated carbon, and even Purigen, Chemipure & "The Poly Filter Pad".
This is a good reason to not use these products as a disease prevention tool (as many mistakenly do with Herbtana and Artemiss), only for occasional treatment use or at least in balance with products such as AAP Wonder Shells. Purigen and especially Chemipure along with "The Poly Filter Pad" are more "medium" oxidizers so extra care should be exercised with their use.
This does NOT mean you should not use Purigen or Poly Pads, just be aware of this important negative side to Redox and counter these with Original AAP Wonder Shells or similar mineral reducers that are added slowly.
Strong/Fast Oxidizers: Potassium Permanganate, Chlorine, Chloramines, Nitric Acid. While the use of Potassium Permanganate in an aquarium for clarifying or flukes control is certainly useful, this product should be very limited for use "in-tank". While Nitric acid is a natural byproduct of bio-filtration, it is still none the less something that can make for a poor Redox Balance rather quickly and is the most common cause of an unhealthy aquarium Redox. Unfortunately, the simple point that Nitric acid is a strong oxidizer is missed by many, including "The Aquarium Wiki" which infers that this is a reducer which is 100% INCORRECT!
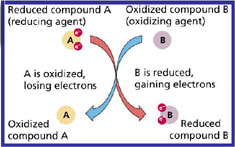
Oxidation describes the loss of an Electropositive or gain of an Electronegative by a molecule, atom or ion. Another way to look at this is to lose, or cause to lose, hydrogen atoms. EXAMPLE: Redox processes such as the oxidation of carbon to yield carbon dioxide. Oxidation is the LACK of electrons by a molecule.
Oxidation is when the molecule can accept electrons from a reduced molecule, thus oxidizing.
Reduction describes the gain of an Electropositive or loss of an Electronegative by a molecule, atom or ion. Another way to look at this is removing oxygen atoms or add hydrogen atoms. EXAMPLE: The reduction of carbon by hydrogen to yield methane (CH4). Reduction is the GAIN of electrons by a molecule. Reduction is when a molecule can give a positive charge to an oxidized molecule, thus ceasing the oxidation of the molecule.
Another example: Calcium (Ca2+) or Magnesium (Mg2+) which initially are composed of two positively charged ions immersed in a sea of movable electrons may have given up all possible electrons to cells/molecules under oxidation. It is for this reason, then that positively charged calcium and magnesium supplies must be constantly renewed; without this “fresh” positively charged calcium, etc. your Redox balance will suffer.
Think of it this way: A battery "works" only when a positive and a negative electrode are present to maintain an electrical current. When the positive plates become exhausted, the battery is no longer functional, even though the metal plates and other "ingredients" for the battery are still present. So it is that your GH or Calcium Test may show adequate minerals, but these minerals have been oxidized and thus rendering the test inaccurate as per ESSENTIAL positively charged calcium ions. This why it is folly to attempt to drive down GH to very low levels in a misguided attempt to replicate certain environmental biotopes based on old school opinions of GH.
Here is an excellent article further explaining how a battery works as it relates to Redox, both oxidation and reduction, and both Anodes and Cathodes (think Anions and Cations). This also explains why a mineral "salt such as CaCO3 (Calcium Carbonate) can have a positive (Cation) ion charge:
• Basics of Electrochemistry (Please note this is a different article as the previous one sadly no longer exists)
What is noteworthy in reading the above article is it speaks of an ionic solution, which our aquarium water is not quite the same as a battery here (saltwater is more so than freshwater), however this also points out the importance of not using totally neutral distilled/de-ionized water to house your fish, as then there is definitely no ionic solution for this current. QUOTE from the old article that is no longer available: "Ions in solution will move from an area of high energy toward an area of low energy producing, a current."
Where the battery comparisons further work, especially for the nay-sayers who claim "we are only measuring ions with a GH test", is that as the calcium Cations are used up, the calcium tends to bind to anything in the aquarium, from objects to even coral in reef aquariums. I have observed this 100s of times (if not 1000s). Similar to this action in a battery. QUOTE: "When the lead acid battery accepts charge, the sulfuric acid gets heavier, causing the specific gravity (SG) to increase. As the SoC decreases through discharge, the sulfuric acid removes itself from the electrolyte and binds to the plate, forming lead sulfate."
Reference:
• How to Measure State-of-charge
It is also noteworthy that while a TDS meter is a good device for any advanced aquarium keeper to own, a TDS meter measures conductivity and pretty much everything that is in the aquarium water, thus not necessarily measuring GH (& KH for other reasons) which COULD be a mistake as reducing Calcium Cations could still be low. If you are really looking to "go pro" definitely own a good TDS Meter (such as the AAP/TMC) ALONG WITH a good ORP/pH Meter (and of course some basic GH & KH test kits too).
Reference:
• Aquarium Chemistry; TDS
Please read on as I will go into further depth as the article progresses, especially as Redox relates to aquatic health.
Even the average aquarist should consider this water parameter when all other parameters check out, yet fish continue to be susceptible to disease. Not only do my years of research and observations with 1000s of aquariums bears out this is an important subject to consider, but growing research in human disease resistance does as well (Redox is constantly discussed in medical research papers, and the higher Redox level previous thought of as healthy have been shown otherwise).
As research grows, knowing what can affect oxidation and reduction which are both important in their own way can help an aquarium keeper deal better with sick fish or an aquarium that has a sudden build up of organics. Even lighting and quality of light is showing to have an affect on Redox based on tests!
However, some in the aquarium keeping community still seem to be in the dark as per this growing documented research.
Redox, also known as Redox Potential, oxidation potential, & ORP (oxidation reduction potential) describes the ability for the loss of an electron by a molecule, atom or ion to the gain of an electron by another molecule, atom or ion. Without this ability to gain electrons, many minerals cannot be absorbed and properly assimilated, especially in times of stress.
So, it is very important to keep a healthy Redox Balance via both sides of the Redox equation: (a) Normal oxidizers such as proper/optimum dissolved oxygen levels. (b) To counter oxidative stress (often artificially induced in our aquariums); via proper positively charged mineral levels (such as Calcium and Magnesium) and even level 1 or higher UV Sterilization.
Often, we as aquarium keepers perform methods to counter oxidative stress without even knowing it, this includes GH boosters used in the "Estimative Index of Dosing" for planted aquariums. In fact, this is how I got started in this research starting in the 1980s, as my observations between my 1000s of client aquariums were showing different results with different aquarium keeping practices, and often these results took months if not years to observe the often subtle but sometimes substantial difference.
Basically we are attempting to provide enough oxidation to provide ample oxygen, allow for biological organic waste breakdown, and not allow too much bacteria in the water column. WHILE AT THE SAME TIME, not add oxidative stress to inhabitants in the aquarium and allow optimum osmoregulation too.
Many in the past have stated exact numbers are the "best" Redox reading for an aquarium (myself included in the past). My research has found this to be incorrect. In some aquariums, a higher Redox of 350 mV may be a good Redox to obtain (for oxidation) when high organics and decomposition is lowering water quality. HOWEVER, for a new or established aquarium that is properly functioning this is not a number you should be necessarily "shooting" for correct Redox balance, as high Redox does not necessarily equate to a healthy aquarium.
A Better simple explanation of "Healthy Redox" is a "Balanced Redox" that recognizes the need for oxidizers to keep clean well oxygenated aquarium water (especially during treatment) while also recognizing that reduction is NECESSARY to balance out oxidizers such as nitric acid that long term affect fish health. So please read on to better understand!
It is also noteworthy that there is a correlation between Redox Potential and pH (Reference: University of Florida; Biochemistry of Wetlands.
Redox also relates to water changes and the amount/frequency thereof. This article has information about using Potassium Permanganate in a very simple (& fun) test to determine the health of your aquarium before and after a water change: Aquarium Cleaning; Reasons (See Part 7, Redox Section).
We should note that we have both slow and fast Redox reducers and slow and fast Redox oxidizers.
Generally in a healthy aquarium regular use of "fast" oxidizers or reducers is not necessary (which the exception of occasional water conditioner use for chlorine reduction). Fast reducers or oxidizers also generally are relatively temporary, although chloramines are definitely a fast reducer that is not temporary.
Examples include:
Slow Reducers: AAP Wonder Shells, use of constant slow flow through water change with mineralized water, some plant ferts, oolitic sand in reaction chamber.
Fast Reducers: Most water conditioners, including Vitamin C or powdered AAP Wonder Shells
Slow (or low level) to Medium Oxidizers: Products such as Methylene Blue (at high concentrations... can be a reducer at low therapeutic levels), Melafix, Herbtana, Artemiss, activated carbon, and even Purigen, Chemipure & "The Poly Filter Pad".
This is a good reason to not use these products as a disease prevention tool (as many mistakenly do with Herbtana and Artemiss), only for occasional treatment use or at least in balance with products such as AAP Wonder Shells. Purigen and especially Chemipure along with "The Poly Filter Pad" are more "medium" oxidizers so extra care should be exercised with their use.
This does NOT mean you should not use Purigen or Poly Pads, just be aware of this important negative side to Redox and counter these with Original AAP Wonder Shells or similar mineral reducers that are added slowly.
Strong/Fast Oxidizers: Potassium Permanganate, Chlorine, Chloramines, Nitric Acid. While the use of Potassium Permanganate in an aquarium for clarifying or flukes control is certainly useful, this product should be very limited for use "in-tank". While Nitric acid is a natural byproduct of bio-filtration, it is still none the less something that can make for a poor Redox Balance rather quickly and is the most common cause of an unhealthy aquarium Redox. Unfortunately, the simple point that Nitric acid is a strong oxidizer is missed by many, including "The Aquarium Wiki" which infers that this is a reducer which is 100% INCORRECT!
Oxidation describes the loss of an Electropositive or gain of an Electronegative by a molecule, atom or ion. Another way to look at this is to lose, or cause to lose, hydrogen atoms. EXAMPLE: Redox processes such as the oxidation of carbon to yield carbon dioxide. Oxidation is the LACK of electrons by a molecule.
Oxidation is when the molecule can accept electrons from a reduced molecule, thus oxidizing.
Reduction describes the gain of an Electropositive or loss of an Electronegative by a molecule, atom or ion. Another way to look at this is removing oxygen atoms or add hydrogen atoms. EXAMPLE: The reduction of carbon by hydrogen to yield methane (CH4). Reduction is the GAIN of electrons by a molecule. Reduction is when a molecule can give a positive charge to an oxidized molecule, thus ceasing the oxidation of the molecule.
Another example: Calcium (Ca2+) or Magnesium (Mg2+) which initially are composed of two positively charged ions immersed in a sea of movable electrons may have given up all possible electrons to cells/molecules under oxidation. It is for this reason, then that positively charged calcium and magnesium supplies must be constantly renewed; without this “fresh” positively charged calcium, etc. your Redox balance will suffer.
Think of it this way: A battery "works" only when a positive and a negative electrode are present to maintain an electrical current. When the positive plates become exhausted, the battery is no longer functional, even though the metal plates and other "ingredients" for the battery are still present. So it is that your GH or Calcium Test may show adequate minerals, but these minerals have been oxidized and thus rendering the test inaccurate as per ESSENTIAL positively charged calcium ions. This why it is folly to attempt to drive down GH to very low levels in a misguided attempt to replicate certain environmental biotopes based on old school opinions of GH.
Here is an excellent article further explaining how a battery works as it relates to Redox, both oxidation and reduction, and both Anodes and Cathodes (think Anions and Cations). This also explains why a mineral "salt such as CaCO3 (Calcium Carbonate) can have a positive (Cation) ion charge:
• Basics of Electrochemistry (Please note this is a different article as the previous one sadly no longer exists)
What is noteworthy in reading the above article is it speaks of an ionic solution, which our aquarium water is not quite the same as a battery here (saltwater is more so than freshwater), however this also points out the importance of not using totally neutral distilled/de-ionized water to house your fish, as then there is definitely no ionic solution for this current. QUOTE from the old article that is no longer available: "Ions in solution will move from an area of high energy toward an area of low energy producing, a current."
Where the battery comparisons further work, especially for the nay-sayers who claim "we are only measuring ions with a GH test", is that as the calcium Cations are used up, the calcium tends to bind to anything in the aquarium, from objects to even coral in reef aquariums. I have observed this 100s of times (if not 1000s). Similar to this action in a battery. QUOTE: "When the lead acid battery accepts charge, the sulfuric acid gets heavier, causing the specific gravity (SG) to increase. As the SoC decreases through discharge, the sulfuric acid removes itself from the electrolyte and binds to the plate, forming lead sulfate."
Reference:
• How to Measure State-of-charge
It is also noteworthy that while a TDS meter is a good device for any advanced aquarium keeper to own, a TDS meter measures conductivity and pretty much everything that is in the aquarium water, thus not necessarily measuring GH (& KH for other reasons) which COULD be a mistake as reducing Calcium Cations could still be low. If you are really looking to "go pro" definitely own a good TDS Meter (such as the AAP/TMC) ALONG WITH a good ORP/pH Meter (and of course some basic GH & KH test kits too).
Reference:
• Aquarium Chemistry; TDS
Please read on as I will go into further depth as the article progresses, especially as Redox relates to aquatic health.
Oxidized Water: Oxidized water with its Redox potential of +700 to +800 mV* is an oxidizing agent that can withdraw electrons from bacteria and kill it. The oxidized water can be used to clean hands, sterilize utensils, and treat minor wounds. This is why products such as Melafix which is an oxidizer (albeit mild), is effective at sterilizing a wound or similar *mV = Millivolts or 1/1000 of a volt of water conductivity.
Here are a few oxidizers: Ozone (O3; Oxidation potential= +2.1), hydrogen peroxide (H2O2; Oxidation potential= +1.82), chlorine (Cl2) and chloramines (NH2Cl).
Further Reference:
• Hydrogen Peroxide; Aquarium Medications Part 3
As well, some aquarium products are also oxidizers such as Potassium Permanganate (sold in products such as "Jungle/Tetra Clear Water") and Mardel Maracyn Oxy. For this reason, both of these products should be used with great caution in your aquarium (although use in established aquariums with a balanced Redox should not be problem when used as directed). Even mild oxidizing aquarium remedies such as Melafix or Herbtana should be used sparingly as overuse WILL CAUSE oxidative stress, as this is what an oxidizer does, it causes stress to tissues! Think stomach acid, an oxidizer, but you sure would not want your other body tissues exposed to it.
Product Resource:
• Maracyn Oxy from AAP
Reduced Water: Reduced Ionized with a Redox Potential of -250 to -350 mV readily donates its electrons to unusual oxygen radicals and blocks the interaction of the active oxygen with normal molecules.
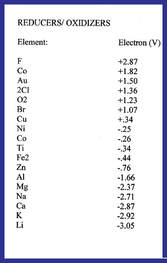
Substances which have the ability to counteract active oxygen by supplying electrons are called scavengers. Reduced water, therefore, can be called scavenging water. Reduced water inhibits excessive fermentation by reducing indirectly metabolites. Please note that the oxidizers have a plus and the reducers have a negative in the chart to the left.
Here are a few reducers, in other words, elements or processes that can transfer electrons to another substance; Magnesium, Calcium, Sodium, and the process of Photosynthesis involves both oxidation and reducing.
As one can see from the graph, elements such as most metals, as well as essential elements for aquatic life: Calcium and Magnesium are major reducers. However, because of this they are also most easily depleted (the elements at the top and the bottom of the graph are most easily depleted in their oxidation or reducing properties).
Most aquarium water conditioners are also Redox Reducers, albeit very short term/temporary reducers. Examples include SeaChem Prime, Tetra/Jungle Start Right, Kordon Novaqua, and many more. As well, Vitamin C which is also used as a method of chlorine removal is a reducer (be aware that Vitamin C used in doses required for chlorine removal in large percentage water changes will drastically drop pH).
Vitamin C is discussed further later in this article #Ref 1.
Further Reference:
• Aquarium Water Conditioners Review & Information
True UVC Sterilization also reduces, by electrically charging (adding an electron) and breaking down oxidizers in the water column. This is clearly demonstrated by picture later in the Aquarium Redox Section of this article. The problem of late is the flood on low-cost UV Sterilizers and/or low output medium pressure UV replacement bulbs sold on Amazon, eBay, and other websites that are at best only capable of clarification and NOT true level one sterilization which can then improve Redox balance (or at the most perform this action poorly).
Aquarium Lighting and the "quality" of this light has also in recent preliminary tests shown to reduce Redox, which has important aquarium keeping implications (both positive and negative, depending upon what is currently happening in an aquarium). See the Aquarium Redox section for more.
Here are a few oxidizers: Ozone (O3; Oxidation potential= +2.1), hydrogen peroxide (H2O2; Oxidation potential= +1.82), chlorine (Cl2) and chloramines (NH2Cl).
Further Reference:
• Hydrogen Peroxide; Aquarium Medications Part 3
As well, some aquarium products are also oxidizers such as Potassium Permanganate (sold in products such as "Jungle/Tetra Clear Water") and Mardel Maracyn Oxy. For this reason, both of these products should be used with great caution in your aquarium (although use in established aquariums with a balanced Redox should not be problem when used as directed). Even mild oxidizing aquarium remedies such as Melafix or Herbtana should be used sparingly as overuse WILL CAUSE oxidative stress, as this is what an oxidizer does, it causes stress to tissues! Think stomach acid, an oxidizer, but you sure would not want your other body tissues exposed to it.
Product Resource:
• Maracyn Oxy from AAP
Reduced Water: Reduced Ionized with a Redox Potential of -250 to -350 mV readily donates its electrons to unusual oxygen radicals and blocks the interaction of the active oxygen with normal molecules.
Substances which have the ability to counteract active oxygen by supplying electrons are called scavengers. Reduced water, therefore, can be called scavenging water. Reduced water inhibits excessive fermentation by reducing indirectly metabolites. Please note that the oxidizers have a plus and the reducers have a negative in the chart to the left.
Here are a few reducers, in other words, elements or processes that can transfer electrons to another substance; Magnesium, Calcium, Sodium, and the process of Photosynthesis involves both oxidation and reducing.
As one can see from the graph, elements such as most metals, as well as essential elements for aquatic life: Calcium and Magnesium are major reducers. However, because of this they are also most easily depleted (the elements at the top and the bottom of the graph are most easily depleted in their oxidation or reducing properties).
Most aquarium water conditioners are also Redox Reducers, albeit very short term/temporary reducers. Examples include SeaChem Prime, Tetra/Jungle Start Right, Kordon Novaqua, and many more. As well, Vitamin C which is also used as a method of chlorine removal is a reducer (be aware that Vitamin C used in doses required for chlorine removal in large percentage water changes will drastically drop pH).
Vitamin C is discussed further later in this article #Ref 1.
Further Reference:
• Aquarium Water Conditioners Review & Information
True UVC Sterilization also reduces, by electrically charging (adding an electron) and breaking down oxidizers in the water column. This is clearly demonstrated by picture later in the Aquarium Redox Section of this article. The problem of late is the flood on low-cost UV Sterilizers and/or low output medium pressure UV replacement bulbs sold on Amazon, eBay, and other websites that are at best only capable of clarification and NOT true level one sterilization which can then improve Redox balance (or at the most perform this action poorly).
Aquarium Lighting and the "quality" of this light has also in recent preliminary tests shown to reduce Redox, which has important aquarium keeping implications (both positive and negative, depending upon what is currently happening in an aquarium). See the Aquarium Redox section for more.
Simple Test to Understand Redox: Take an 8 oz. glass of water, then add enough Potassium Permanganate to change the color to a mild red, then take any aquarium water conditioner such as Prime or Start Right and slowly add drops to the water and watch the red be "reduced" (neutralized) to a clear brown color. What you are observing is the reduction of an oxidizer (potassium Permanganate) by a Reducer (generally Sodium Thiosulfate is used in these conditioners).
Product Resources:
• Potassium Permanganate (Jungle Clear Water)
• SeaChem Prime
For an advanced Redox/pH tester:
• Professional Waterproof Portable pH/ORP Meter
This picture/graphic below explains some basics of Redox:
Product Resources:
• Potassium Permanganate (Jungle Clear Water)
• SeaChem Prime
For an advanced Redox/pH tester:
• Professional Waterproof Portable pH/ORP Meter
This picture/graphic below explains some basics of Redox:
An important note, is that although oxidation is a necessary part of biochemistry for fish and all animals (such as for energy production), the NORMAL HEALTHY state is one of reduction.
During normal biochemical processes molecules that are normally reducers give up their electrons (in much the same way a car battery does until re-charged), so without a recharging via the addition of new minerals that are high in these electrons or even processes such as UVC sterilization (or even high PAR lighting), your aquatic biochemistry will suffer and eventually so will your fish!
Further References:
• UV Sterilization; Complete Facts & Information
• Aquarium Lighting; PAR
One more basic generalization to consider before moving into more depth is this: Water that is of low pH (acid), in general, measures high ORP while water of high pH (alkaline) measures low ORP (please note that this is a broad generalization). However, in natural water (generally spring water), acidity of minus ions and alkalinity of plus ions can coexist (more about this in Natural Redox).
Also worthy of note, a 2012 human research has shown that an intake of 75% alkaline foods versus 25% acid foods are best for essential Redox Balance. While this is not a perfect comparison, sine the human body changes the pH of food as it progresses from the stomach into the small intestine, this can still be reasonably extrapolated to fish in the water environment and foods should allow for this as well since the physiology of fish and humans are similar. As well since fish live in their liquid environment, water parameters such as Redox have a more definite effect on fish than with humans. This of course often confounds those who constantly dump copious amounts of acid buffers or similar in their aquarium or worry about a generally fictitious "too high GH".
During normal biochemical processes molecules that are normally reducers give up their electrons (in much the same way a car battery does until re-charged), so without a recharging via the addition of new minerals that are high in these electrons or even processes such as UVC sterilization (or even high PAR lighting), your aquatic biochemistry will suffer and eventually so will your fish!
Further References:
• UV Sterilization; Complete Facts & Information
• Aquarium Lighting; PAR
One more basic generalization to consider before moving into more depth is this: Water that is of low pH (acid), in general, measures high ORP while water of high pH (alkaline) measures low ORP (please note that this is a broad generalization). However, in natural water (generally spring water), acidity of minus ions and alkalinity of plus ions can coexist (more about this in Natural Redox).
Also worthy of note, a 2012 human research has shown that an intake of 75% alkaline foods versus 25% acid foods are best for essential Redox Balance. While this is not a perfect comparison, sine the human body changes the pH of food as it progresses from the stomach into the small intestine, this can still be reasonably extrapolated to fish in the water environment and foods should allow for this as well since the physiology of fish and humans are similar. As well since fish live in their liquid environment, water parameters such as Redox have a more definite effect on fish than with humans. This of course often confounds those who constantly dump copious amounts of acid buffers or similar in their aquarium or worry about a generally fictitious "too high GH".
Relative Hydrogen (rH)
This section on rH compliments of Rogier Van Vlissingen. This takes ORP reading a step further as to showing the true health of the water.
The rH/Relative Hydrogen Score, is a better measure of whether the water is actually oxidizing or reducing than ORP is. ORP is a proxy, rH directly measures atomic H in the water, (which includes H-), so it is still not a direct measure of H-, but it's closer than ORP.
In 1997 Prof. Sanetaka Shirahata of Kyoto University published a paper in BBRC (a peer reviewed journal, Biochemical and Biophysical Research Communications), which demonstrated for the first time that H- persisted in electrolyzed water, putting to rest an industry myth that the reason for the health effects of the alkaline water from electrolysis was the very alkalinity of it. Not so, it was the presence of H-, which science up to that point had believed could not exist but for nano seconds.
Dr. Hidemitsu Hayashi pointed out that in several natural bodies of water the same thing was found, and he began manufacturing his mineral sticks, which were a much cheaper and more effective way to create HRW, hydrogen rich water. As per my history section later in this article, it was in part the use of Cation rich mineral blocks that I myself stumbled upon this concept after observing improved fish health.
FURTHER (Quote from The Relative Hydrogen Score, aka the rH Score): "The rH scale employs the same logarithmic Bar scale used to express gas pressure in terms of order of magnitude, and the rH scale runs from 0 to 42; 28 is mid-point (balance), below 28 is reducing, above 28 oxidizing... Therefore, an rH score of 0 would indicate maximum possible reducing (antioxidant) power, while a score of 42 would indicate the maximum possible oxidizing environment."
So, rH yields a hydrogen proton-unbiased measure of absolute reducing potential of a substance, eliminating effects due to pH. rH is log1/hydrogen partial pressure; a one-unit change in rH indicates a 10X change. E.g., a shift on the Barr scale of rH from a score of 27 to one of 26 indicates a 10X increase in reducing power. rH 28 = H partial pressure (pp) of 10^-28 atmospheres, an rH 11 = H pp is 10^-11 atmospheres, and a 0 rH is the rH value of pure hydrogen at STP. Again, as noted above, an rH score of 0 would indicate maximum possible reducing (aka “antioxidant”) power, while a score of 42 would indicate the maximum possible oxidizing environment.
Nonetheless, ORP and better, rH, have found some degree of acceptance for many years in the fields of high-end aquarium keeping, wine and beer brewing, food storage and food safety as an indicator or relative antioxidant ability, and, since the mid-to-late 90's; in some sectors of the nutritional antioxidant field as an indicator of the same as well. It is somewhat important to note that ORP and rH scores have also been used to some extent for many years in the groundwater and waste water remediation fields as well as measures of relative oxidation or reducing ability of water.
By keeping track of your measurements with a spreadsheet, you can track rH. Below 28 on the scale is reducing, above 28 is oxidizing... therefore any rH below 28 indicates antioxidant effectiveness of the water by the presence of atomic H, which will include H-. We are beginning to find healthy aquariums will have rH reading is between 23 & 28 (although as yet an exact number has not been established for marine aquariums, I suspect it is likely 25-30).
You can use a spreadsheet to do the work. The most exact formula is rH=((ORP+205)/29.58) +2*pH), in simplified form it is ((ORP+200)/30) +(2*pH), but I see no reason not to use the accurate formula.
Further Reading/Reference:
• Everything Aquatic; Redox Results. ORP, PH, rH
• Water Institute, and Munenori KAWAMURA, M.D., Kyowa Medical Clinic
A quote from the above article: "Since May ’85 we have confirmed thousands of clinical improvements, obtained solely by exchanging drinking (as well as cooking water) from tap water to reduced water." Examples of RH Scores Calculated from Various ORP & pH Values.
It is important to note that Aquarium Redox can be a complex subject with some basic principles to also understand, however reading one section of this article will yield incomplete information. For this reason I recommend reading the whole article (as well as links provided) for a more thorough understanding. It may take more than one reading as well.
The rH/Relative Hydrogen Score, is a better measure of whether the water is actually oxidizing or reducing than ORP is. ORP is a proxy, rH directly measures atomic H in the water, (which includes H-), so it is still not a direct measure of H-, but it's closer than ORP.
In 1997 Prof. Sanetaka Shirahata of Kyoto University published a paper in BBRC (a peer reviewed journal, Biochemical and Biophysical Research Communications), which demonstrated for the first time that H- persisted in electrolyzed water, putting to rest an industry myth that the reason for the health effects of the alkaline water from electrolysis was the very alkalinity of it. Not so, it was the presence of H-, which science up to that point had believed could not exist but for nano seconds.
Dr. Hidemitsu Hayashi pointed out that in several natural bodies of water the same thing was found, and he began manufacturing his mineral sticks, which were a much cheaper and more effective way to create HRW, hydrogen rich water. As per my history section later in this article, it was in part the use of Cation rich mineral blocks that I myself stumbled upon this concept after observing improved fish health.
FURTHER (Quote from The Relative Hydrogen Score, aka the rH Score): "The rH scale employs the same logarithmic Bar scale used to express gas pressure in terms of order of magnitude, and the rH scale runs from 0 to 42; 28 is mid-point (balance), below 28 is reducing, above 28 oxidizing... Therefore, an rH score of 0 would indicate maximum possible reducing (antioxidant) power, while a score of 42 would indicate the maximum possible oxidizing environment."
So, rH yields a hydrogen proton-unbiased measure of absolute reducing potential of a substance, eliminating effects due to pH. rH is log1/hydrogen partial pressure; a one-unit change in rH indicates a 10X change. E.g., a shift on the Barr scale of rH from a score of 27 to one of 26 indicates a 10X increase in reducing power. rH 28 = H partial pressure (pp) of 10^-28 atmospheres, an rH 11 = H pp is 10^-11 atmospheres, and a 0 rH is the rH value of pure hydrogen at STP. Again, as noted above, an rH score of 0 would indicate maximum possible reducing (aka “antioxidant”) power, while a score of 42 would indicate the maximum possible oxidizing environment.
Nonetheless, ORP and better, rH, have found some degree of acceptance for many years in the fields of high-end aquarium keeping, wine and beer brewing, food storage and food safety as an indicator or relative antioxidant ability, and, since the mid-to-late 90's; in some sectors of the nutritional antioxidant field as an indicator of the same as well. It is somewhat important to note that ORP and rH scores have also been used to some extent for many years in the groundwater and waste water remediation fields as well as measures of relative oxidation or reducing ability of water.
By keeping track of your measurements with a spreadsheet, you can track rH. Below 28 on the scale is reducing, above 28 is oxidizing... therefore any rH below 28 indicates antioxidant effectiveness of the water by the presence of atomic H, which will include H-. We are beginning to find healthy aquariums will have rH reading is between 23 & 28 (although as yet an exact number has not been established for marine aquariums, I suspect it is likely 25-30).
You can use a spreadsheet to do the work. The most exact formula is rH=((ORP+205)/29.58) +2*pH), in simplified form it is ((ORP+200)/30) +(2*pH), but I see no reason not to use the accurate formula.
Further Reading/Reference:
• Everything Aquatic; Redox Results. ORP, PH, rH
• Water Institute, and Munenori KAWAMURA, M.D., Kyowa Medical Clinic
A quote from the above article: "Since May ’85 we have confirmed thousands of clinical improvements, obtained solely by exchanging drinking (as well as cooking water) from tap water to reduced water." Examples of RH Scores Calculated from Various ORP & pH Values.
It is important to note that Aquarium Redox can be a complex subject with some basic principles to also understand, however reading one section of this article will yield incomplete information. For this reason I recommend reading the whole article (as well as links provided) for a more thorough understanding. It may take more than one reading as well.
Background
My research and interest in Redox goes back to my curiosity as to why certain aquarium maintenance methods were more effective in keeping healthy fish, and the common knowledge of the day did not fully explain results I was achieving.
This article is an out-growth of many 1000s of observations and some controlled tests in search of an answer/explanation that commonly held aquarium keeping believes simply could not explain or were even contradictory to (such as commonly held views about GH in freshwater aquariums as well as the effect of UVC on all aquariums as per disease resistance). As I research this subject more and more, I find that it answers many of these results, but I am still learning too as I am already finding out more to Redox than just mineral Cations and oxidation versus reduction such as Relative Hydrogen (rH).
I base this article on Redox studies as it corresponds to humans and all fish, animals, and even plants.
I have found from practical experience and research that often a lot more can be learned from medical studies or other university level studies than from many aquarium articles and sadly some otherwise excellent forums (these articles/forums usually just take bits from another article without any real research, often with old ideas being simply regurgitated).
Originally my statement on Redox was: "I do not believe this is a subject that many aquarists should stress over", but a lot has been learned about this subject since I first started researching this in the 1980s. The more I have studied this subject as well as the newest research shows that all aquarists should have at least a basic understanding of this subject as it has major implications for fish and aquatic health that often go against commonly held aquarium keeping beliefs.
I have also changed my opinions about this subject based on newer evidence about a balanced Redox.
One such opinion was "should you have a positive or reducing Redox". I based my earlier opinion on a higher Redox of +300 mV based on many studies of ocean waters and simply repeating what I was told, but newer human research and my own tests over the last 15 plus years have lead me to where I am now, that a BALANCED Redox is important.
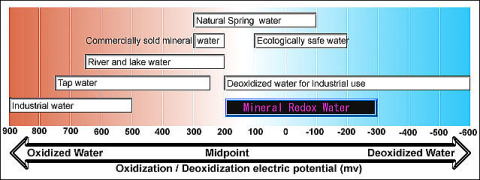
What is a balanced Redox? The best research indicates a Redox that can respond to both the oxidation and reduction needs of the environment and more importantly the fish or other aquarium inhabitant. This can be a reading between +300 to -200 mV (a higher Redox is sometimes needed when oxidation is necessary due to high organic de-composition). See the chart below (ecologically safe water):
This article is an out-growth of many 1000s of observations and some controlled tests in search of an answer/explanation that commonly held aquarium keeping believes simply could not explain or were even contradictory to (such as commonly held views about GH in freshwater aquariums as well as the effect of UVC on all aquariums as per disease resistance). As I research this subject more and more, I find that it answers many of these results, but I am still learning too as I am already finding out more to Redox than just mineral Cations and oxidation versus reduction such as Relative Hydrogen (rH).
I base this article on Redox studies as it corresponds to humans and all fish, animals, and even plants.
I have found from practical experience and research that often a lot more can be learned from medical studies or other university level studies than from many aquarium articles and sadly some otherwise excellent forums (these articles/forums usually just take bits from another article without any real research, often with old ideas being simply regurgitated).
Originally my statement on Redox was: "I do not believe this is a subject that many aquarists should stress over", but a lot has been learned about this subject since I first started researching this in the 1980s. The more I have studied this subject as well as the newest research shows that all aquarists should have at least a basic understanding of this subject as it has major implications for fish and aquatic health that often go against commonly held aquarium keeping beliefs.
I have also changed my opinions about this subject based on newer evidence about a balanced Redox.
One such opinion was "should you have a positive or reducing Redox". I based my earlier opinion on a higher Redox of +300 mV based on many studies of ocean waters and simply repeating what I was told, but newer human research and my own tests over the last 15 plus years have lead me to where I am now, that a BALANCED Redox is important.
What is a balanced Redox? The best research indicates a Redox that can respond to both the oxidation and reduction needs of the environment and more importantly the fish or other aquarium inhabitant. This can be a reading between +300 to -200 mV (a higher Redox is sometimes needed when oxidation is necessary due to high organic de-composition). See the chart below (ecologically safe water):
Maintaining a proper Redox Balance (or Potential) which includes the reducing side of the equation is a lot like having an antioxidant vitamin in the aquarium water (although a more accurate description would be magnetically charged water). The Aquarium Redox is also just one more parameter towards good disease prevention and long-term fish health and growth.
Please see this article about Aquarium Disease prevention:
• Aquarium Disease Prevention
Looking at the oxidizing side of the Redox equation; Bleach and Potassium Permanganate are oxidizers (Potassium Permanganate is often used for disease treatment and water clarification in ponds and aquariums). However, you would not want your fish living in a constantly oxidizing environment for long term fish health, rather you would want your fish in a balanced redox environment which includes a Reducing environment where free radicals are not damaging the cells of your fish and causing problems with Osmoregulation. An example would be HLLE in marine fish where evidence points to a direct link of an oxidizing environment without a constant input of Redox reduction to balance this being a causative factor.
Further Information:
• Fish Osmoregulation
An oxidizing environment is what you get with a positive Redox of generally +350 mV and is OK (& necessary) for certain periods such as during disease treatments, however you would not want to keep your fish in constant medication any more than in a constant strongly oxidizing environment (PLEASE read the section about Natural Redox including the quote and outside Redox for a better understanding of this concept)!
Another IMPORTANT consideration is a healthy Redox affects the electrolyte balance in your aquarium. Calcium as well as Magnesium are both important reducers when positively charged, however they are both easily oxidized as well (please see the chart in the Redox Potential Basics section). So, for proper reduction, these elements, or at least their positive ion charge need to be replaced on a constant basis (read further in this article for more on this subject).
The Redox Potential explains a lot of the results I was getting as the fish have a better ability to resist a disease sometimes simply by having a healthier immune system and osmoregulation. This pertains to a Balanced Redox potential (which includes a proper magnetic charge in the water) that I have found a properly installed and constructed UV Sterilizer aids in maintaining as well as proper mineralization of reducers and constant replenishment of these minerals, such as calcium, and regular water changes. that lower nitric acid and DOC levels (dissolved organic compounds).
Further Reference:
• Aquarium Cleaning, reasons, methods
What got me started in researching this topic was that I noticed and documented, differences in health, vitality and especially disease resistance within different aquariums among the literally 100s (if not 1000s) of contract customer aquariums I kept going back to 1978. I noted a difference in health with very regular small, mineralized water changes or flow through water change methods (including ponds), UV sterilizers, and added minerals/ electrolytes (such as Wonder Shells or similar methods/products). This improvement/difference was noted over tanks kept with more traditional methods such as "Amazon" biotopes where RO water was used, and no replacement mineralization (Cation replacement) was used on an ongoing basis.
The observations first began in earnest at the Bahooka Restaurant in 1978 where I had over 110 aquariums, mostly over 80 gallons, and all with low bio loads, the same feeding schedule, the same exact filtration method: However, variables in in water changes & use of ongoing mineral replacements.
Later starting in 1992 at another client with multiple aquariums, I attempted more controlled tests with 6 tanks that also included UVC as a variable based on many single aquarium observations of where one tank would have a UV Sterilizer, UV clarifier (not level one), or none at all. These tests consisted of "fancy" goldfish aquariums. The variables beyond the control tank included feeding, water changes (using tap water with high mineral content), ongoing constant use of mineral replacement blocks, level one capable UV sterilizers. This also included introduction of new fish at the same intervals.
The goldfish had fewer instances of general diseases as well as infestations such as Ich, Anchor Worms, and other problems with the variables of constant replenishment of mineral cations, regular water changes, the use of correctly installed efficient UV sterilizers (all of which contribute to balanced Redox).
Best results included use of all variables, but each of these variables produced better results than the aquarium with none of these variables put into practice (as was noted n my 100s of more anecdotal observations to this point).
What is/was noteworthy is that the UV Sterilizers were not even run within the parameters that would kill Ich outright, yet I still was observing lower instances of Ich!!! WHY? The answer is simple in that the aquarium had a healthier Redox Balance that aided in the fish' own natural immunity and resistance to Ich!
PLEASE NOTE: Many UV Sterilizers now marketed for under $50 CANNOT perform effective Redox reduction due to their poor dwell time, low-cost medium pressure UV bulbs, and other factors, these are at best clarifiers and should NEVER be purchased unless that is all you desire from your UV! And to critics who push cheap Amazon and eBay UV clarifiers: Yes, I have had extensive experience in UV Sterilizer/Clarifier use and YES, I've both witnessed a difference in level one UV Sterilization in both disease resistance and lowering of oxidative stress (which includes longevity of fish)!!!
Further Reference:
• Actual UV-C Emission from a UV Bulb
This shows that the health of aquarium fish can be directly aided by positive mineral ions/magnetic charge of the water and a properly installed "true" UV Sterilizer, not just by the killing of potential disease pathogens, but also by the effect UVC has on the Redox Balance of an aquarium as per UV Sterilization's affect upon oxidizers in the water column (although ORP readings are NOT changed much by UV Sterilization).
I have been observing many aquariums of "ordinary" fish, both with and without UV Sterilizers (keep in mind that some UVs are useless by design and installation), and the goldfish with every other factor equal (such as mineralization and water changes) that had a UV Sterilizer installed, were generally much healthier.
A High Dwell Time UV Sterilizer works similar to the ozone layer in our upper atmosphere (except in reverse); although the UVC emitted by the sterilizer is itself an oxidizer, the interaction of the UVC inside the unit with oxidizers such as ozone is such that the UV Sterilizer REDUCES these oxidizers and free radicals as well as potentially magnetically charges the water. This was confirmed by Dr. Mamoon Kundi in an email he sent me as per his work using UVC to balance Redox in human studies, and thus improving immune function.
Product Resource:
• AAP/TMC Vecton & Advantage "High Dwell Time" UV Sterilizers
For those interested in checking this out, simply add potassium Permanganate into a tank with a UV Sterilizer and one without. The tank with the UV Sterilizer clears MUCH quicker! If your aquarium has a high bio load, the addition of a properly installed UV Sterilizer can actually temporarily add too much Redox Reduction resulting in a darkening of water color until the Aquarium Redox balances again.
Please Reference:
• Aquarium Bio Load
Unfortunately, the aspect of Redox Balance and Ultraviolet Sterilization is often missed my many aquarists, and sadly many otherwise well-informed reef keepers.
Further Reference:
• UVC Sterilization/Sterilizers and the effect on a Healthy Redox
In further research I noted that a proper Redox Potential improves the health of humans AND fish. A Redox Potential in the -200-400 mV range in human studies has been shown to have the same affect as anti-oxidant preparations as Vitamin C, and many others.
In medical studies, the enhanced oxidizing environment can facilitate the binding of pathogens or antigens to effector cells (a type of lymphocyte that are actively engaged in secreting antibodies) leading to a hyper-responsive innate immune system. Previous work has shown that an oxidizing environment leads to enhanced release of super-oxide and nitric oxide, activation and translocation of nuclear transcription factor and enhanced production of cytokines (proteins and peptides that are signaling compounds produced by animal cells to communicate with one another).
The creation of a reduced environment by addition of antioxidants blunts all of the above primary responses of the innate immune system. My many observations in both fresh & saltwater lend to this principle too as to how fish, both freshwater & marine respond both short & long term to oxidizers and/or the lack of a constant source of reduction to balance out oxidative stress.
Back to my many tests with goldfish tanks over several years in the 1990s; I ran these tests with many different filters and combinations there of. Cleanings were consistent every two weeks with about 25% changes of water via a Gravel Vacuum. Feeding was performed at the same intervals with the same food (at that time Hikari and Spirulina 20), and all the tanks had the same number of goldfish at as close to the same size as possible.
I found the tanks with the Under Gravel Filters performed the worst (and not just in Redox) due to their tendency for DOC (Dissolved Organic Compound) buildup which results in high amounts of Nitric Acid production. UG Filters performed better when combined with a UV Sterilizer and another filter such as a canister filter, however the tanks that performed the best did not have UG filters, rather a combination of two filters (generally a Sponge Filter or Fluidized Filter were part of this combination) AND a UV Sterilizer.
Product Resources:
• Premium Patented AAP Aquarium Sponge Filters
• Premium 3rd generation Fluidized Sand Bed Filters
Some of the tanks were maintained with added minerals/electrolytes, some were not. The best results as per long term health were achieved where minerals cations (positive mineral ions) were added on a constant basis vs. methods that required weekly or similar dosing requirements or non-minerals at all.
That said, please read the rest of the article before I over sell any one on a UV Sterilizer, Mineral Blocks (such as AAP Wonder Shells), or drip mineral applicators (products such as SeaChem Replenish can be mixed with water and dripped into the aquarium). As often good aquatic husbandry is a major factor in a healthy Redox Potential (balance).
Another interesting aspect of Redox potential is the correlation of a certain Redox level and the growth of Blue Green Algae (Cyanobacteria) in Aquariums and lakes. This is an on-going study however Redox does SEEM to play some role in the aquarium and lake “plague”.
For more information about this, I recommend reading this article:
• Blue Green Algae (Cyanobacteria) in Aquariums; what it is and how to control it.
Currently this is just an opinion/theory though.
Please see this article about Aquarium Disease prevention:
• Aquarium Disease Prevention
Looking at the oxidizing side of the Redox equation; Bleach and Potassium Permanganate are oxidizers (Potassium Permanganate is often used for disease treatment and water clarification in ponds and aquariums). However, you would not want your fish living in a constantly oxidizing environment for long term fish health, rather you would want your fish in a balanced redox environment which includes a Reducing environment where free radicals are not damaging the cells of your fish and causing problems with Osmoregulation. An example would be HLLE in marine fish where evidence points to a direct link of an oxidizing environment without a constant input of Redox reduction to balance this being a causative factor.
Further Information:
• Fish Osmoregulation
An oxidizing environment is what you get with a positive Redox of generally +350 mV and is OK (& necessary) for certain periods such as during disease treatments, however you would not want to keep your fish in constant medication any more than in a constant strongly oxidizing environment (PLEASE read the section about Natural Redox including the quote and outside Redox for a better understanding of this concept)!
Another IMPORTANT consideration is a healthy Redox affects the electrolyte balance in your aquarium. Calcium as well as Magnesium are both important reducers when positively charged, however they are both easily oxidized as well (please see the chart in the Redox Potential Basics section). So, for proper reduction, these elements, or at least their positive ion charge need to be replaced on a constant basis (read further in this article for more on this subject).
The Redox Potential explains a lot of the results I was getting as the fish have a better ability to resist a disease sometimes simply by having a healthier immune system and osmoregulation. This pertains to a Balanced Redox potential (which includes a proper magnetic charge in the water) that I have found a properly installed and constructed UV Sterilizer aids in maintaining as well as proper mineralization of reducers and constant replenishment of these minerals, such as calcium, and regular water changes. that lower nitric acid and DOC levels (dissolved organic compounds).
Further Reference:
• Aquarium Cleaning, reasons, methods
What got me started in researching this topic was that I noticed and documented, differences in health, vitality and especially disease resistance within different aquariums among the literally 100s (if not 1000s) of contract customer aquariums I kept going back to 1978. I noted a difference in health with very regular small, mineralized water changes or flow through water change methods (including ponds), UV sterilizers, and added minerals/ electrolytes (such as Wonder Shells or similar methods/products). This improvement/difference was noted over tanks kept with more traditional methods such as "Amazon" biotopes where RO water was used, and no replacement mineralization (Cation replacement) was used on an ongoing basis.
The observations first began in earnest at the Bahooka Restaurant in 1978 where I had over 110 aquariums, mostly over 80 gallons, and all with low bio loads, the same feeding schedule, the same exact filtration method: However, variables in in water changes & use of ongoing mineral replacements.
Later starting in 1992 at another client with multiple aquariums, I attempted more controlled tests with 6 tanks that also included UVC as a variable based on many single aquarium observations of where one tank would have a UV Sterilizer, UV clarifier (not level one), or none at all. These tests consisted of "fancy" goldfish aquariums. The variables beyond the control tank included feeding, water changes (using tap water with high mineral content), ongoing constant use of mineral replacement blocks, level one capable UV sterilizers. This also included introduction of new fish at the same intervals.
The goldfish had fewer instances of general diseases as well as infestations such as Ich, Anchor Worms, and other problems with the variables of constant replenishment of mineral cations, regular water changes, the use of correctly installed efficient UV sterilizers (all of which contribute to balanced Redox).
Best results included use of all variables, but each of these variables produced better results than the aquarium with none of these variables put into practice (as was noted n my 100s of more anecdotal observations to this point).
What is/was noteworthy is that the UV Sterilizers were not even run within the parameters that would kill Ich outright, yet I still was observing lower instances of Ich!!! WHY? The answer is simple in that the aquarium had a healthier Redox Balance that aided in the fish' own natural immunity and resistance to Ich!
PLEASE NOTE: Many UV Sterilizers now marketed for under $50 CANNOT perform effective Redox reduction due to their poor dwell time, low-cost medium pressure UV bulbs, and other factors, these are at best clarifiers and should NEVER be purchased unless that is all you desire from your UV! And to critics who push cheap Amazon and eBay UV clarifiers: Yes, I have had extensive experience in UV Sterilizer/Clarifier use and YES, I've both witnessed a difference in level one UV Sterilization in both disease resistance and lowering of oxidative stress (which includes longevity of fish)!!!
Further Reference:
• Actual UV-C Emission from a UV Bulb
This shows that the health of aquarium fish can be directly aided by positive mineral ions/magnetic charge of the water and a properly installed "true" UV Sterilizer, not just by the killing of potential disease pathogens, but also by the effect UVC has on the Redox Balance of an aquarium as per UV Sterilization's affect upon oxidizers in the water column (although ORP readings are NOT changed much by UV Sterilization).
I have been observing many aquariums of "ordinary" fish, both with and without UV Sterilizers (keep in mind that some UVs are useless by design and installation), and the goldfish with every other factor equal (such as mineralization and water changes) that had a UV Sterilizer installed, were generally much healthier.
A High Dwell Time UV Sterilizer works similar to the ozone layer in our upper atmosphere (except in reverse); although the UVC emitted by the sterilizer is itself an oxidizer, the interaction of the UVC inside the unit with oxidizers such as ozone is such that the UV Sterilizer REDUCES these oxidizers and free radicals as well as potentially magnetically charges the water. This was confirmed by Dr. Mamoon Kundi in an email he sent me as per his work using UVC to balance Redox in human studies, and thus improving immune function.
Product Resource:
• AAP/TMC Vecton & Advantage "High Dwell Time" UV Sterilizers
For those interested in checking this out, simply add potassium Permanganate into a tank with a UV Sterilizer and one without. The tank with the UV Sterilizer clears MUCH quicker! If your aquarium has a high bio load, the addition of a properly installed UV Sterilizer can actually temporarily add too much Redox Reduction resulting in a darkening of water color until the Aquarium Redox balances again.
Please Reference:
• Aquarium Bio Load
Unfortunately, the aspect of Redox Balance and Ultraviolet Sterilization is often missed my many aquarists, and sadly many otherwise well-informed reef keepers.
Further Reference:
• UVC Sterilization/Sterilizers and the effect on a Healthy Redox
In further research I noted that a proper Redox Potential improves the health of humans AND fish. A Redox Potential in the -200-400 mV range in human studies has been shown to have the same affect as anti-oxidant preparations as Vitamin C, and many others.
In medical studies, the enhanced oxidizing environment can facilitate the binding of pathogens or antigens to effector cells (a type of lymphocyte that are actively engaged in secreting antibodies) leading to a hyper-responsive innate immune system. Previous work has shown that an oxidizing environment leads to enhanced release of super-oxide and nitric oxide, activation and translocation of nuclear transcription factor and enhanced production of cytokines (proteins and peptides that are signaling compounds produced by animal cells to communicate with one another).
The creation of a reduced environment by addition of antioxidants blunts all of the above primary responses of the innate immune system. My many observations in both fresh & saltwater lend to this principle too as to how fish, both freshwater & marine respond both short & long term to oxidizers and/or the lack of a constant source of reduction to balance out oxidative stress.
Back to my many tests with goldfish tanks over several years in the 1990s; I ran these tests with many different filters and combinations there of. Cleanings were consistent every two weeks with about 25% changes of water via a Gravel Vacuum. Feeding was performed at the same intervals with the same food (at that time Hikari and Spirulina 20), and all the tanks had the same number of goldfish at as close to the same size as possible.
I found the tanks with the Under Gravel Filters performed the worst (and not just in Redox) due to their tendency for DOC (Dissolved Organic Compound) buildup which results in high amounts of Nitric Acid production. UG Filters performed better when combined with a UV Sterilizer and another filter such as a canister filter, however the tanks that performed the best did not have UG filters, rather a combination of two filters (generally a Sponge Filter or Fluidized Filter were part of this combination) AND a UV Sterilizer.
Product Resources:
• Premium Patented AAP Aquarium Sponge Filters
• Premium 3rd generation Fluidized Sand Bed Filters
Some of the tanks were maintained with added minerals/electrolytes, some were not. The best results as per long term health were achieved where minerals cations (positive mineral ions) were added on a constant basis vs. methods that required weekly or similar dosing requirements or non-minerals at all.
That said, please read the rest of the article before I over sell any one on a UV Sterilizer, Mineral Blocks (such as AAP Wonder Shells), or drip mineral applicators (products such as SeaChem Replenish can be mixed with water and dripped into the aquarium). As often good aquatic husbandry is a major factor in a healthy Redox Potential (balance).
Another interesting aspect of Redox potential is the correlation of a certain Redox level and the growth of Blue Green Algae (Cyanobacteria) in Aquariums and lakes. This is an on-going study however Redox does SEEM to play some role in the aquarium and lake “plague”.
For more information about this, I recommend reading this article:
• Blue Green Algae (Cyanobacteria) in Aquariums; what it is and how to control it.
Currently this is just an opinion/theory though.
Natural Redox
Over geologic time, abiogenic, biogenic, and anthropogenic deposits were accumulated in rivers, lakes and seas.
TERMS:
• Abiogenic = not biological in origin.
• Biogenic = produced by living organisms.
• Anthropogenic = processes that are derived from human activities.
Simultaneously all three of these processes proceed to self-purify natural water on the basis of the chemical reactions of oxidation and reduction. Living organisms such as fish are adapted to NOT "perfectly clean water", but to ecologically clean water with definite content of organic and inorganic compounds, micro-admixtures, Magnetized ions, and even bacteria or saprophytes (saprophytes are an organism that grows on and derives its metabolic energy from dead or decaying organic matter, such as most fungi). The range of mentioned components of ecologically clean water is the integral result of oxidation and reduction self-purification of water.
At the initial stages of this natural cycle, toxic hydrophobic organic compounds (molecules that are repelled from a mass of water) are oxidized to the hydrophilic form, which is a molecule, or portion of a molecule, that is typically charge-polarized and capable of hydrogen bonding, enabling it to dissolve more readily in water than in oil or other hydrophobic solvents. These are characterized by the better biological compatibility.
Energy of oxidation by way of degrading organic compounds is absorbed during reduction chemical transformations. Excess ions of the heavy elements transform into insoluble, inert, nontoxic forms. The concentration of ions of light elements are then stabilized. Water organisms, live in such water, and land animals drink it.
Spring waters, which are considered to be the most clean, according to the ecological criteria, are filtered through the rocks and subjected to the oxidation and reduction, sorption and catalytic influence.
As a rule, they are mineralized and include non-volatile organic substances, which are detected by the permanganate oxidability (expressed by oxidability by potassium permanganate and potassium dichromate). Put more bluntly; only water that has been naturally filtered by percolation deep into the earth, heated under pressure to super steam at the mantle and re-imprinted by the clays, gravels and minerals that it passes by on its way to the surface at 4C is fully mature and full of useful electromagnetic energy.
ORP can change rapidly, and Redox potentials above 400 mV long term are dangerous to life. Good quality water for life is lower than 350mV, and water of lower than 100mV is effective for disease healing purpose when the part of body is particularly oxidized (harmed) by disease (although generally speaking for out aquarium/pond purposes we would not want our Redox this low long term as oxidation is an important aspect of Redox balance).
In tests using electrochemical purification, water keeps its initial neutral values of pH, but ORP (Oxidation Reduction Potential) of water shifts toward the negative (electrode or reduction) values. The resulting purified water obtains the antioxidant properties with characteristics of pharmacological activity close to the properties of the antioxidant preparations (a -tocopherol, b -carotene, vitamin C etc.). In cases of dilution of poly-vitamin preparations in electrochemically purified water, the ORP of mixtures is decreased by approximately 200 - 400 mV in relation to the control solutions with non-treated drinking water.
The above partially explains how a proper Redox Potential improves the health of the fish or other aquatic organisms as I have observed. Bringing this back to my observations of goldfish aquariums; All aquariums with the same filtration (canister filter with sponge filter), the same feeding schedule and food, even the same basic goldfish (a mix of Ryukins and Orandas); The aquariums with the quality, properly installed UV Sterilizers (again assuming proper dissolved oxygen levels) not only had less incidence of disease, but general health, appetite, vigor, growth, and water clarity were all improved.
Here is a Quote about pH vs. Redox: Traditionally we have judged the properties of water from the standpoint of pH, in other words whether water is acidic or alkaline. According to Dr. Yoshiaki Matsuo PhD., the inventor of the Ionized Water unit: "In my opinion, Redox potential is more important than pH. The importance of pH is over emphasized. For example, the average pH of blood is 7.4 and acidosis or alkalosis are defined according to deviation within the range of 7.4 +- 0.005. But nothing has been discussed about ORP, or oxidation-reduction potential."
A further point on human studies shows this: “Dr. Uchiyama, working at a general hospital in Japan, conducted a complete medical checkup with his 871 clients. The result was 92 had normal results (judged healthy) and 279 requiring further consultation or treatment for their health (judged potentially sick).
He measured OPR of all of their urine. It was found that many healthy clients showed ORP voltage of +50 to +74 and clients judged unhealthy showed +100 to +124. Healthy persons tended to have lower voltage of ORP, less oxidation potential in their body.”
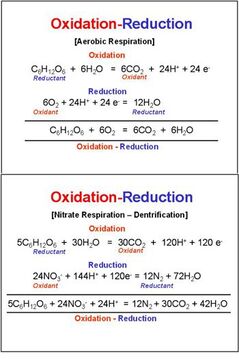
Below is a chart that shows Redox Reactions that support life:
(From https://www.esf.edu/efb/schulz/Limnology/redox.html, which is now sadly gone as well.)
TERMS:
• Abiogenic = not biological in origin.
• Biogenic = produced by living organisms.
• Anthropogenic = processes that are derived from human activities.
Simultaneously all three of these processes proceed to self-purify natural water on the basis of the chemical reactions of oxidation and reduction. Living organisms such as fish are adapted to NOT "perfectly clean water", but to ecologically clean water with definite content of organic and inorganic compounds, micro-admixtures, Magnetized ions, and even bacteria or saprophytes (saprophytes are an organism that grows on and derives its metabolic energy from dead or decaying organic matter, such as most fungi). The range of mentioned components of ecologically clean water is the integral result of oxidation and reduction self-purification of water.
At the initial stages of this natural cycle, toxic hydrophobic organic compounds (molecules that are repelled from a mass of water) are oxidized to the hydrophilic form, which is a molecule, or portion of a molecule, that is typically charge-polarized and capable of hydrogen bonding, enabling it to dissolve more readily in water than in oil or other hydrophobic solvents. These are characterized by the better biological compatibility.
Energy of oxidation by way of degrading organic compounds is absorbed during reduction chemical transformations. Excess ions of the heavy elements transform into insoluble, inert, nontoxic forms. The concentration of ions of light elements are then stabilized. Water organisms, live in such water, and land animals drink it.
Spring waters, which are considered to be the most clean, according to the ecological criteria, are filtered through the rocks and subjected to the oxidation and reduction, sorption and catalytic influence.
As a rule, they are mineralized and include non-volatile organic substances, which are detected by the permanganate oxidability (expressed by oxidability by potassium permanganate and potassium dichromate). Put more bluntly; only water that has been naturally filtered by percolation deep into the earth, heated under pressure to super steam at the mantle and re-imprinted by the clays, gravels and minerals that it passes by on its way to the surface at 4C is fully mature and full of useful electromagnetic energy.
ORP can change rapidly, and Redox potentials above 400 mV long term are dangerous to life. Good quality water for life is lower than 350mV, and water of lower than 100mV is effective for disease healing purpose when the part of body is particularly oxidized (harmed) by disease (although generally speaking for out aquarium/pond purposes we would not want our Redox this low long term as oxidation is an important aspect of Redox balance).
In tests using electrochemical purification, water keeps its initial neutral values of pH, but ORP (Oxidation Reduction Potential) of water shifts toward the negative (electrode or reduction) values. The resulting purified water obtains the antioxidant properties with characteristics of pharmacological activity close to the properties of the antioxidant preparations (a -tocopherol, b -carotene, vitamin C etc.). In cases of dilution of poly-vitamin preparations in electrochemically purified water, the ORP of mixtures is decreased by approximately 200 - 400 mV in relation to the control solutions with non-treated drinking water.
The above partially explains how a proper Redox Potential improves the health of the fish or other aquatic organisms as I have observed. Bringing this back to my observations of goldfish aquariums; All aquariums with the same filtration (canister filter with sponge filter), the same feeding schedule and food, even the same basic goldfish (a mix of Ryukins and Orandas); The aquariums with the quality, properly installed UV Sterilizers (again assuming proper dissolved oxygen levels) not only had less incidence of disease, but general health, appetite, vigor, growth, and water clarity were all improved.
Here is a Quote about pH vs. Redox: Traditionally we have judged the properties of water from the standpoint of pH, in other words whether water is acidic or alkaline. According to Dr. Yoshiaki Matsuo PhD., the inventor of the Ionized Water unit: "In my opinion, Redox potential is more important than pH. The importance of pH is over emphasized. For example, the average pH of blood is 7.4 and acidosis or alkalosis are defined according to deviation within the range of 7.4 +- 0.005. But nothing has been discussed about ORP, or oxidation-reduction potential."
A further point on human studies shows this: “Dr. Uchiyama, working at a general hospital in Japan, conducted a complete medical checkup with his 871 clients. The result was 92 had normal results (judged healthy) and 279 requiring further consultation or treatment for their health (judged potentially sick).
He measured OPR of all of their urine. It was found that many healthy clients showed ORP voltage of +50 to +74 and clients judged unhealthy showed +100 to +124. Healthy persons tended to have lower voltage of ORP, less oxidation potential in their body.”
Below is a chart that shows Redox Reactions that support life:
(From https://www.esf.edu/efb/schulz/Limnology/redox.html, which is now sadly gone as well.)
Balance
I have mostly discussed Redox Reduction and its misunderstood importance. However, there does need to be a balance between reduction and oxidation for proper chemistry within a fish or most all living animal organisms.
Cellular redox balance is critical for maintaining normal function and preventing damage to DNA and proteins, which may lead to accelerated aging and more.
While it is generally recognized that preserving cellular redox balance and preventing a high concentration of reactive oxygen species (ROS, i.e., free radicals) is crucial to maintaining healthy organ and systems in living organisms, scientists are still debating the specific species responsible for maintaining this balance.
A new discovery published in 2018 is about biologically-active sulfur species known as persulfides (R–SSH). Persulfides were initially believed to only form on proteins as a means of easily modulating the protein’s activity. However, recent research has demonstrated that the same enzymes responsible for producing H2S produce free persulfides enzymatically in cells.
What makes persulfides particularly interesting is the wide variety of chemical reactions they can partake in, owing to the ability of sulfur to occupy eight oxidation states. Previous studies on persulfide chemistry, while limited, have demonstrated that persulfides have a greater reducing potential (greater antioxidant behavior) than most other sulfur species identified in cells. Taken altogether, our knowledge that persulfides are generated enzymatically in cells, combined with their enhanced reducing potential, indicates that persulfides may be key players in maintaining cellular redox balance.
Where this might fit in with aquarium keeping might be very interesting, as healthy reduction to balance out oxidation is often what is missing in our aquariums (even among advanced aquarium keepers unaware of the importance of Redox Balance) for true long-term health of aquatic inhabitants.
For the more scientific minded, the diagram/picture below explains how persulfides affect Redox Balance:
Cellular redox balance is critical for maintaining normal function and preventing damage to DNA and proteins, which may lead to accelerated aging and more.
While it is generally recognized that preserving cellular redox balance and preventing a high concentration of reactive oxygen species (ROS, i.e., free radicals) is crucial to maintaining healthy organ and systems in living organisms, scientists are still debating the specific species responsible for maintaining this balance.
A new discovery published in 2018 is about biologically-active sulfur species known as persulfides (R–SSH). Persulfides were initially believed to only form on proteins as a means of easily modulating the protein’s activity. However, recent research has demonstrated that the same enzymes responsible for producing H2S produce free persulfides enzymatically in cells.
What makes persulfides particularly interesting is the wide variety of chemical reactions they can partake in, owing to the ability of sulfur to occupy eight oxidation states. Previous studies on persulfide chemistry, while limited, have demonstrated that persulfides have a greater reducing potential (greater antioxidant behavior) than most other sulfur species identified in cells. Taken altogether, our knowledge that persulfides are generated enzymatically in cells, combined with their enhanced reducing potential, indicates that persulfides may be key players in maintaining cellular redox balance.
Where this might fit in with aquarium keeping might be very interesting, as healthy reduction to balance out oxidation is often what is missing in our aquariums (even among advanced aquarium keepers unaware of the importance of Redox Balance) for true long-term health of aquatic inhabitants.
For the more scientific minded, the diagram/picture below explains how persulfides affect Redox Balance:
Some of the above referenced/copied from:
• On-Demand Release Of Persulfides As Antioxidants
Biochemistry as it relates to Redox Balance: There are enzymes within a body that are reactive oxygen species (ROS) and others that are reactive nitrogen species (RNS). A disturbance in the oxidation–reduction state of the cell, in which ROS production exceeds antioxidant defenses, is called oxidative stress. By analogy, nitrosative stress is an impairment in nitric oxide (NO) signaling caused by increased amounts of RNS, which may be caused by, or associated with, a disturbance in the Redox state. As discussed earlier, free radicals are highly reactive molecules with unpaired electrons.
Free radical chemistry is the underpinning of 2 broad classes of signaling molecules in biological systems: ROS, which are reactive intermediates of oxygen metabolism, and a closely related group of RNS. The forms of ROS that are relevant in biological systems include the superoxide radical (O2•–), hydrogen peroxide (H2O2), and hydroxyl radical (OH•). RNS of biological importance include NO, low- and high-molecular-weight S-nitrosothiols, and peroxynitrite (ONOO–).
Superoxide and other ROS are produced in the mitochondria during oxidative phosphorylation as a normal byproduct of aerobic respiration in fish (and other animals). Superoxide is formed by the 1-electron reduction of molecular oxygen, resulting in a free radical. However, ROS are not intrinsically destructive; on the contrary, increasing evidence shows that they play necessary roles in normal signal transduction.
In low concentrations, they are implicated as second messengers primarily through inhibition of phosphatases, acting downstream of effectors such as platelet-derived growth factor, epidermal growth factor, tumor necrosis factor agonists, and interleukin. In higher concentrations, however, they take on pathophysiological roles. ROS affect the oxidative modification of diverse molecules, including DNA, proteins, lipids, and sugars, potentially leading to toxicity.
The problem begins when either ROS (oxidation) or RNS (Reduction) get out of balance and unfortunately in closed aquarium or pond systems this is all too common. An aquarium with poor circulation and low oxygen levels can swing towards poor oxidation, while more commonly Reduction is halted by the fish’ body inability to reduce these superoxides.
Reduction is dependant on production of RNS; NOS3 (endothelial NOS) are found in a variety of cell types and are regulated by binding to calcium and calmodulin (calcium-binding protein expressed in all eukaryotic cells). NOS2 (inducible NOS), on the other hand, has very high baseline affinity for calcium and calmodulin; therefore, its activity is effectively independent of calcium concentration. The importance of calcium in this reaction is often missed by most aquarists based on the many emails I get and comments I read on many misinformed aquatic forums.
As well, the reduction side of this equation can also be interrupted by the oxidation of other reducing chemicals such as ascorbate (vitamin C) and reduced thiols, especially glutathione (a tripeptide). These reduced chemicals become temporarily oxidized when they interact (giving up of their free electrons) with oxidizing agents and oxidizing free radicals.
Further Reference:
• About Glutathione
A more basic aspect of Redox Balance is the water we use can have a direct impact as well. The water we use, whether from the tap or Reverse Osmosis/De-ionized this too can affect your Redox Balance, sometimes negatively. Generally speaking, tap water if only altered with chlorine or chloramines may or may not have the necessary mineral cations to bring a poor Redox back into balance.
The use of RO or DI water, while a very practical way to improve and control water parameters (especially with soft water freshwater species or reef aquaria), one should note that the necessary mineral positive mineral ions (cations) are usually gone (removed). The resulting excess oxygen and more acid water is often way too high of an oxidizing environment. For this reason, it is often important to re-minerals these water sources with products such as SeaChem Replenish or Wonder Shells.
Product Resource:
• TMC Aquarium Reverse Osmosis Water Filter System
• On-Demand Release Of Persulfides As Antioxidants
Biochemistry as it relates to Redox Balance: There are enzymes within a body that are reactive oxygen species (ROS) and others that are reactive nitrogen species (RNS). A disturbance in the oxidation–reduction state of the cell, in which ROS production exceeds antioxidant defenses, is called oxidative stress. By analogy, nitrosative stress is an impairment in nitric oxide (NO) signaling caused by increased amounts of RNS, which may be caused by, or associated with, a disturbance in the Redox state. As discussed earlier, free radicals are highly reactive molecules with unpaired electrons.
Free radical chemistry is the underpinning of 2 broad classes of signaling molecules in biological systems: ROS, which are reactive intermediates of oxygen metabolism, and a closely related group of RNS. The forms of ROS that are relevant in biological systems include the superoxide radical (O2•–), hydrogen peroxide (H2O2), and hydroxyl radical (OH•). RNS of biological importance include NO, low- and high-molecular-weight S-nitrosothiols, and peroxynitrite (ONOO–).
Superoxide and other ROS are produced in the mitochondria during oxidative phosphorylation as a normal byproduct of aerobic respiration in fish (and other animals). Superoxide is formed by the 1-electron reduction of molecular oxygen, resulting in a free radical. However, ROS are not intrinsically destructive; on the contrary, increasing evidence shows that they play necessary roles in normal signal transduction.
In low concentrations, they are implicated as second messengers primarily through inhibition of phosphatases, acting downstream of effectors such as platelet-derived growth factor, epidermal growth factor, tumor necrosis factor agonists, and interleukin. In higher concentrations, however, they take on pathophysiological roles. ROS affect the oxidative modification of diverse molecules, including DNA, proteins, lipids, and sugars, potentially leading to toxicity.
The problem begins when either ROS (oxidation) or RNS (Reduction) get out of balance and unfortunately in closed aquarium or pond systems this is all too common. An aquarium with poor circulation and low oxygen levels can swing towards poor oxidation, while more commonly Reduction is halted by the fish’ body inability to reduce these superoxides.
Reduction is dependant on production of RNS; NOS3 (endothelial NOS) are found in a variety of cell types and are regulated by binding to calcium and calmodulin (calcium-binding protein expressed in all eukaryotic cells). NOS2 (inducible NOS), on the other hand, has very high baseline affinity for calcium and calmodulin; therefore, its activity is effectively independent of calcium concentration. The importance of calcium in this reaction is often missed by most aquarists based on the many emails I get and comments I read on many misinformed aquatic forums.
As well, the reduction side of this equation can also be interrupted by the oxidation of other reducing chemicals such as ascorbate (vitamin C) and reduced thiols, especially glutathione (a tripeptide). These reduced chemicals become temporarily oxidized when they interact (giving up of their free electrons) with oxidizing agents and oxidizing free radicals.
Further Reference:
• About Glutathione
A more basic aspect of Redox Balance is the water we use can have a direct impact as well. The water we use, whether from the tap or Reverse Osmosis/De-ionized this too can affect your Redox Balance, sometimes negatively. Generally speaking, tap water if only altered with chlorine or chloramines may or may not have the necessary mineral cations to bring a poor Redox back into balance.
The use of RO or DI water, while a very practical way to improve and control water parameters (especially with soft water freshwater species or reef aquaria), one should note that the necessary mineral positive mineral ions (cations) are usually gone (removed). The resulting excess oxygen and more acid water is often way too high of an oxidizing environment. For this reason, it is often important to re-minerals these water sources with products such as SeaChem Replenish or Wonder Shells.
Product Resource:
• TMC Aquarium Reverse Osmosis Water Filter System
Redox as it's to Aquariums
My continued research into aquarium Redox does not provide exact numbers, but I would refer back tot the numbers I refer to in the rH section. Moreover, I would also refer back to this point from the overview section of this article: Basically, we are attempting to provide enough oxidation to provide ample oxygen, allow for biological organic waste breakdown, and not allow too much bacteria in the water column. WHILE AT THE SAME TIME, not add oxidative stress to inhabitants in the aquarium and allow optimum osmoregulation too.
The point being, Redox Potential is often in “flux”, so I personally believe following healthy aquatic husbandry methods (that consider the affect on Redox) is an aquarists best bet in maintaining this balance. This includes a higher Redox (as high as +350) when oxidation is necessary due to high organic decomposition as well as lower numbers for long term lower oxidative stress.
There are multiple mechanisms for maintaining a good redox balance from a good oxygen exchange for oxidation to mineral ions and certain light energy for important reduction.
Another way to look at the Redox Potential in aquariums is to the relation of waste particles and acid production, as well as dissolved oxygen and most importantly depletion of key important elements. The more waste, the less Redox Potential (ORP) due to more nitric acid production and the depletion of key elements, but this is an oversimplification as there is much more to it than this.
The point being, Redox Potential is often in “flux”, so I personally believe following healthy aquatic husbandry methods (that consider the affect on Redox) is an aquarists best bet in maintaining this balance. This includes a higher Redox (as high as +350) when oxidation is necessary due to high organic decomposition as well as lower numbers for long term lower oxidative stress.
There are multiple mechanisms for maintaining a good redox balance from a good oxygen exchange for oxidation to mineral ions and certain light energy for important reduction.
Another way to look at the Redox Potential in aquariums is to the relation of waste particles and acid production, as well as dissolved oxygen and most importantly depletion of key important elements. The more waste, the less Redox Potential (ORP) due to more nitric acid production and the depletion of key elements, but this is an oversimplification as there is much more to it than this.
You really need to look at the electrolyte balance in your aquarium. Calcium, as well as Magnesium, are both important reducers, however they are both easily oxidized as well (see the graph in the Redox Basics section). As well the use of oxidizers that "scrub" your aquarium such as Purigen and Chemipure can throw off this balance. So, for proper balance, which includes reduction, these elements or more importantly their "electrical charge" (positive ions/cations) need to be replaced!
Obviously high organic wastes also require an input of oxidation (higher Redox for Redox balance), but this should not be ongoing due to a poor balance of reducing to oxidation because of the lack of redox reducing mechanisms as well as a bio load the aquariums system cannot support. This is good reason to not overstock and aquarium, as there is a point whereby it is impossible to balance your aquarium Redox other than for short term.
The nitrogen cycle requires an input of oxidation, but it also requires an input of reduction due to the excess oxidizers/free electrons (Anions) produced. Without this balance, one creates an environment where the fish could therefore be attacked by these Anions (although a planted aquarium may not suffer from this aspect as much since many plants utilize these nutrients prior to entering the nitrogen cycle). This is also the problem of a high bio load in an aquarium or pond. This fact is borne out in both my long-term observations and tests. The picture above displays this process.
Reference:
• University of Florida; Biochemistry of Wetlands
SIMPLY PUT, THESE FACTORS HAVE THE MOST AFFECT IN REDOX BALANCE MAINTENANCE.
Water Changes: This is the most obvious and simple, however this is often not sufficient and sometimes the new water used does not have adequate mineral ions (especially if RO water is used even in part), so supplementing with mineral replenishers, such as SeaChem Replenish, Wonder Shells, etc. (even during water changes may be necessary).
PLEASE NOTE that performing inadequate water changes or worse, only topping off water for evaporation does not adequately replenish important redox reducing ions of calcium or magnesium. Most any aquarium is going to utilize these ions at a far higher rate than simply adding “fresh” water or even some water changes will ever replenish. The end result of such poor maintenance procedures will result in climbing GH readings, yet your aquarium (or pond) ability to maintain a healthy Redox balance will suffer.
Product Resources:
• SeaChem Replenish
• Wonder Shells; Unique FRESH Version ONLY from AAP
Addition of positive mineral ions in between water changes, especially during times of stress or high bio loads can increase the Redox Reduction to counter oxidative effects on fish. Use of products or methods (such as AAP Wonder Shells) to add positive mineral ions are considered "slow" reducers as these do not have large initial impact upon Redox unlike fast reducers such as any water conditioners (such as AAP Prime).
It is noteworthy that despite attacks by websites with obviously little experience in professional aquarium keeping (such as aquariumscience.org), that the Calcium ions (Cations) are indeed reducing agents and that my experiments and experience over the years have shown that product such as AAP Wonder Shells lose this Cation "charge" over time and why the purchase of fresh Wonder Shells is essential for full benefits (and yes, my experience has shown that indeed oxidative stress affects fish as well as other life).
For planted aquarium keepers, the regular use of many ferts such as NilocG Macros/Micros will have a Redox reduction too, and will thus render products such AAP Wonder Shells basically inert/unnecessary. However, a slow dissolving product such as an AAP Wonder Shell or SeaLab will have more impact during a time of stress than a product added in intervals (a level one UV Sterilizer will also act more quickly on oxidizers in the water column too).
Lack of positive mineral ions during stress can be a factor in a Columnaris outbreak or failure of treatment. Use of products that can further degrade Redox Balance can be even more detrimental to treatment or prevention of Columnaris. Examples include "The Poly Pad", "Chemi-Pure", and even AAP Purigen.
Tests with a Redox Meter showed a 20% drop in Redox (ORP) when mixing in shavings from an AAP Wonder Shell using a control aquarium water sampling both before and after.
As noted earlier in this article, I am not saying use of Purigen or Poly Pads should be avoided, just to be aware of the negative effect on Redox balance, especially with fish that might already be under stress from disease, transfer, etc. and use measures such as constant replenishing mineral Cation blocks such as the original AAP Wonder Shell. I've also found that when using these oxidizing products in a planted aquarium, that the normal ferts do not over come this negative side affect very well.
Further Reference:
• Aquarium Columnaris
Use of a water conditioner such as AAP Prime, Start Right, etc. and especially SeaChem/AAP StressGuard. These conditioners are immediate or "Fast" Redox reducers having a very immediate, but also very short-term impact upon Redox. An example would be how the use of the water conditioner "Prime" will immediately remove any chlorine (an oxidizer) while a slow reducer such as a Wonder Shell will take hours to do the same. Because of this large immediate, albeit temporary impact on Redox, it is not a good idea to keep adding water conditioners daily such as in an attempt to control ammonia (NH3) as this can affect the "oxidizing" nitrogen cycle.
StressGuard is a very unique product that addresses oxidative and other stress causes in fish with proven/tested results by many professionals. This is an excellent product to often use instead of medications for fish that are struggling with health.
Pictures and further information about StressGaurd can be found here:
• Aquarium Disease Prevention; Recommended Products to have on hand
Product Resource:
• AAP/SeaChem StressGuard (please support this free information website with your purchase from AAP)
Further Reference/Reading:
• Aquarium Water Conditioners; Review, Information
Obviously high organic wastes also require an input of oxidation (higher Redox for Redox balance), but this should not be ongoing due to a poor balance of reducing to oxidation because of the lack of redox reducing mechanisms as well as a bio load the aquariums system cannot support. This is good reason to not overstock and aquarium, as there is a point whereby it is impossible to balance your aquarium Redox other than for short term.
The nitrogen cycle requires an input of oxidation, but it also requires an input of reduction due to the excess oxidizers/free electrons (Anions) produced. Without this balance, one creates an environment where the fish could therefore be attacked by these Anions (although a planted aquarium may not suffer from this aspect as much since many plants utilize these nutrients prior to entering the nitrogen cycle). This is also the problem of a high bio load in an aquarium or pond. This fact is borne out in both my long-term observations and tests. The picture above displays this process.
Reference:
• University of Florida; Biochemistry of Wetlands
SIMPLY PUT, THESE FACTORS HAVE THE MOST AFFECT IN REDOX BALANCE MAINTENANCE.
Water Changes: This is the most obvious and simple, however this is often not sufficient and sometimes the new water used does not have adequate mineral ions (especially if RO water is used even in part), so supplementing with mineral replenishers, such as SeaChem Replenish, Wonder Shells, etc. (even during water changes may be necessary).
PLEASE NOTE that performing inadequate water changes or worse, only topping off water for evaporation does not adequately replenish important redox reducing ions of calcium or magnesium. Most any aquarium is going to utilize these ions at a far higher rate than simply adding “fresh” water or even some water changes will ever replenish. The end result of such poor maintenance procedures will result in climbing GH readings, yet your aquarium (or pond) ability to maintain a healthy Redox balance will suffer.
Product Resources:
• SeaChem Replenish
• Wonder Shells; Unique FRESH Version ONLY from AAP
Addition of positive mineral ions in between water changes, especially during times of stress or high bio loads can increase the Redox Reduction to counter oxidative effects on fish. Use of products or methods (such as AAP Wonder Shells) to add positive mineral ions are considered "slow" reducers as these do not have large initial impact upon Redox unlike fast reducers such as any water conditioners (such as AAP Prime).
It is noteworthy that despite attacks by websites with obviously little experience in professional aquarium keeping (such as aquariumscience.org), that the Calcium ions (Cations) are indeed reducing agents and that my experiments and experience over the years have shown that product such as AAP Wonder Shells lose this Cation "charge" over time and why the purchase of fresh Wonder Shells is essential for full benefits (and yes, my experience has shown that indeed oxidative stress affects fish as well as other life).
For planted aquarium keepers, the regular use of many ferts such as NilocG Macros/Micros will have a Redox reduction too, and will thus render products such AAP Wonder Shells basically inert/unnecessary. However, a slow dissolving product such as an AAP Wonder Shell or SeaLab will have more impact during a time of stress than a product added in intervals (a level one UV Sterilizer will also act more quickly on oxidizers in the water column too).
Lack of positive mineral ions during stress can be a factor in a Columnaris outbreak or failure of treatment. Use of products that can further degrade Redox Balance can be even more detrimental to treatment or prevention of Columnaris. Examples include "The Poly Pad", "Chemi-Pure", and even AAP Purigen.
Tests with a Redox Meter showed a 20% drop in Redox (ORP) when mixing in shavings from an AAP Wonder Shell using a control aquarium water sampling both before and after.
As noted earlier in this article, I am not saying use of Purigen or Poly Pads should be avoided, just to be aware of the negative effect on Redox balance, especially with fish that might already be under stress from disease, transfer, etc. and use measures such as constant replenishing mineral Cation blocks such as the original AAP Wonder Shell. I've also found that when using these oxidizing products in a planted aquarium, that the normal ferts do not over come this negative side affect very well.
Further Reference:
• Aquarium Columnaris
Use of a water conditioner such as AAP Prime, Start Right, etc. and especially SeaChem/AAP StressGuard. These conditioners are immediate or "Fast" Redox reducers having a very immediate, but also very short-term impact upon Redox. An example would be how the use of the water conditioner "Prime" will immediately remove any chlorine (an oxidizer) while a slow reducer such as a Wonder Shell will take hours to do the same. Because of this large immediate, albeit temporary impact on Redox, it is not a good idea to keep adding water conditioners daily such as in an attempt to control ammonia (NH3) as this can affect the "oxidizing" nitrogen cycle.
StressGuard is a very unique product that addresses oxidative and other stress causes in fish with proven/tested results by many professionals. This is an excellent product to often use instead of medications for fish that are struggling with health.
Pictures and further information about StressGaurd can be found here:
• Aquarium Disease Prevention; Recommended Products to have on hand
Product Resource:
• AAP/SeaChem StressGuard (please support this free information website with your purchase from AAP)
Further Reference/Reading:
• Aquarium Water Conditioners; Review, Information
Use of a UV Sterilizer which impacts Redox Balance in a different way than water changes or additional minerals: The UVC irradiation destroys destructive oxidizers and aids in establishing the proper balance of electromagnetism of the water column which can otherwise add oxidative stress to fish. This in turn improves a fish' immune response to diseases and other stressors in the water.
The picture above graphically displays the dramatic difference. In the control tank (left) and tank with a level one capable UVC Sterilizer (AAP 7-Watt Compact UV), potassium permanganate (which is a strong oxidizer) was added at the same rate. Potassium Permanganate reduces from a red color to brown to clear. It is very obvious the difference in the two tanks, which means that there is less oxidative stress to fish. Thus, this increases the fish' ability fight off diseases in particular diseases such as Lateral Line disease in marine aquariums which seem to be more prevalent when oxidative stress is higher.
While there is some criticism of UV Sterilizer use claiming if used to the point of killing or altering all potential pathogens, this would be detrimental similar to never exposing your children to anything and keeping them isolated in a bubble. This is only correct in that the immune system should not be sheltered from every possibly malady so as to be able to provide a healthy immune response when needed.
However, the complete destruction of all pathogens is not what happens with correct UV Sterilizer use.
First this is not possible for even the best UV to destroy all pathogens in an aquarium or pond (or altering pathogens making them unable to multiply) as for one not all pathogens are in the water column, many or already opportunistic living in the fish, substrate, or elsewhere.
What a well-designed and properly installed true UV Sterilizer can do is improve immune function via improved Redox Balance. This aspect of a UV Sterilizer is so often missed or even utilized by those who own a true UV Sterilizer but only run a UV after a problem has occurred. This is similar to how one might give their children Echinacea before sending them off to school since Echinacea improves immune response, but in itself generally does not destroy disease pathogens.
However, it is noteworthy that the use of Ozone Generators does NOT accomplish the same results with Redox balance, despite similar results in sterilization (I found this stated in some very confused articles and forum posts I have read).
Product Resource:
• Aquarium/Pond UV Sterilizers CAPABLE of improving Redox
Lighting and the "quality" of this light also impact aquarium Redox. By quality I mean the nanometer wavelengths make a difference on the impact of Redox, with lighting in a more natural "noon time sun" 6500K appearing to have the greatest impact overall. This impact is a reduction of ORP from the time the lights go on in the morning into the day, going back up after lights are off.
The way the redox value changes with light is due to direct molecular bond changes from photon impact. The result of this is an atom/molecule with a higher or lower oxidation state, which is then shown as a change in measured redox value. It's a small but important distinction. Redox is a chemical reaction, while irradiation is direct manipulation of the bonds without a chemical reaction.
The implication is that if you need a higher ORP for oxidation due to organic waste buildup, lights should be reduced. However generally speaking and if your fish are sick, reducing Redox (ORP) is what you want and stronger and better lighting (such as produced by the AquaRay GroBeams and AquaBeams).
Recently reviewed and updated tests have also shown the positive benefit of near infrared to infrared light in the improvement of Redox Reduction and wound healing. Again, this is now a proven fact that has aquatic implications for good lighting that hits these nanometer spikes. Generally, within 5500K to 6700K lighting, although specific 'Red' lighting can help too, such as the TMC Flexi-Red LEDs or TMC HO Colour Plus LEDs.
Product Resources:
• TMC Aquarium LED Lights; Including Colour Plus
See:
• Wise and limited use of Ozoneizers/Ozone Generators (Red Light Therapy)
• Aquarium Lighting; Facts & Information
Important: What is often missed, is that although you may test and, for instance find, high calcium levels in an aquarium via a GH over 300, this does not necessarily mean you have any Redox reduction as I should point out that very basic fact of Redox reduction is the donor (such as calcium) transfers electrons to another substance, and is thus, oxidized itself. This is VERY key, meaning your GH test (or Calcium, Magnesium, etc tests in saltwater) may tell you that you have ample or even too much calcium. HOWEVER, these calcium molecules may have already given up their electrons to other molecules such as fish cell molecules so as to achieve a state of balance in aquarium Redox.
This again is where many will claim that their calcium or other minerals are high enough or even too high (as many still get INCORRECTLY stressed about the high GH brought on by AAP Wonder Shells, totally missing that these mineral blocks are constantly reducing the water via their donation of electrons!).
This is also where water changes and UV Sterilization also help as these processes constantly balance out Redox. Since water changes (which also aid in oxidation, which is the other side of the equation) can only be done in intervals. Unless you have a flow through system, the use of mineral blocks, or other water conditioners as well as UV Sterilization to keep a steadier flow of electrolyte donation is important.
With the above statements in mind, one must look at two VERY basic organic chemical reactions that occur in an aquarium: As stated in the opening section of this article; “oxidation of carbon to yield carbon dioxide”, this describes the process of nitrification or in other words the breakdown of organic wastes by Autotrophic nitrifying bacteria into ammonia and nitrites.
The higher the bio load, the more of this process is occurring. So, it is imperative to replace elements such as negatively charged ions of calcium, magnesium, etc. that are oxidized due to this process.
This is where many aquarists “miss the boat” as per reasons for water changes, replenishing minerals, or other Redox Reduction processes that will balance this out. This is also why tanks without good, ionized mineralization, UV Sterilization, water changes, etc. will often be MUCH less healthy as per disease resistance.
Reduction as also stated in the opening section: “reduction of carbon by hydrogen to yield methane (CH4)”, this describes the de-nitrification of nitrate as it reduces to nitrogen. This process is more common in Marine tanks but can take place in freshwater in certain places where methane production is not an issue such as filters with anaerobic filtration that allow methane to escape (volcanic rock & SeaChem Matrix provide a good place for this process). Also, the growth of plants, especially with strong roots in a sandy substrate allows for de-nitrification/Redox reduction. This is an area where Veggie Filters in Ponds shine in their ability to maintain excellent bio parameters. This also to a lesser degree shows where some green algae growth is not all that bad.
Further Reference:
• Pond Veggie/Bog Filters
All this said, Reduction generally de-nitrification processes CANNOT keep up with oxidation processes necessary for breakdown of organic wastes, which is why help is needed via water changes, mineralization, UV Sterilization, etc. This is where many naysayers of products such as Wonder Shells, or UV Sterilizers TOTALLY miss the point as to how much they can improve water quality. This is also how the use of products such as Volcanic Rock or SeaChem Matrix can also indirectly affect Redox.
Product Resources:
• Volcanic Rock Filter Media, Nitrifying, De-nitrifying
• SeaChem Matrix; Biological Nitrate Remover, De-nitrifier
Important! Put another way, there is certainly a time and place for a high Redox (oxidation) as lower oxygen levels from many causes such as high organic decomposition is generally not desirable, and this will lower your Redox oxidation side of the equation. This is not what I recommend as a means of a reducing Redox environment. However, to not have the proper reducers or creating a highly oxidative environment is also not good for long term fish health.
The Redox Potential is always going to be in flux and as, yet no scientific equilibrium has been established here. Maintaining reducers such as Calcium and Magnesium in your aquarium is just as important as proper levels of dissolved oxygen (which raises Redox). There is evidence both anecdotal and controlled that proper levels of calcium and other antioxidants such as Vitamin C (along with other healthy water parameters) will prevent and even cure Hole in the Head disease (HITH). Redox is an important parameter here.
Further Reference:
• Aquarium Answers, about Hole in the Head Disease
As stated earlier, Redox can be more important than pH, there are definitely correlations to pH and the Redox effect on pH. But you cannot make the assumption that if you have a pH of 8.0, that you have good reduction. Just like with GH, where your GH is high, you can still have low pH. The bottom line is that there are relations between Redox, GH, pH, and even kH, but these are still individual water parameters.
Also as noted in the beginning of this article, this is not a parameter to stress over, HOWEVER, Redox reduction should NOT be blown off as many anecdotal/uninformed commentators do in some aquatic forums. The more I have studied and observed in this subject, the more I feel it should not be ignored!
Effect on Fish: Active oxygen molecules, or free radicals, are produced in the water and the fish’ body. They are extremely reactive and can also attach themselves to normal, healthy cells and damage them genetically. These active oxygen radicals steal electrons from normal, healthy biological molecules.
This electron theft by active oxygen oxidizes tissue and can cause disease; these are proven facts!
This why the constant hammering by some un-informed aquarists (such a popular planted aquarium guru) against UVs, too high a GH (which is not always a good indicator of electrolyte balance anyway), and even the use of mineral blocks (such as Wonder Shells) is ludicrous! Your GH may test higher than desired but in actuality is maintaining a healthy Reducing Redox via the reducing properties of positively charged calcium, magnesium, etc.
Human studies show a clear link between the Redox level in the blood of a healthy person and an unhealthy person, especially when it comes to cancer. Those with cancer and other chronic illnesses show more oxidation. For fish, this means that while oxidizing agents have their place, we should not be regularly dumping oxidizing products in our aquariums, even if they are so-called "natural" products whether it be Microbe-Lift Herbtana or API Melafix. The scientific facts are that over the long term the regular use of these products WILL SHORTEN the life of your fish and make them more susceptible to diseases, including tumors.
Reference:
• Tissue redox activity as a hallmark of carcinogenesis
In short, oxidative stress most definitely plays a role in both short- & long-term fish health and ability to heal. Diseases such as Columnaris and HLLE can be linked to oxidative stress from both my 1000s of observations as well as research.
Effect on plants: In a controlled test conducted with terrestrial plants; plants treated with magnetized water (basically Redox Balanced Water) reacted in a test, with a growth in the order of 20% to 40% bigger than the plants treated with regular water. While no test has been conducted with planted aquariums, it is not rocket science to draw conclusions from this study and apply this to a planted freshwater aquarium and the effect of Redox Balance on plant growth!
Simple Redox Test: A simple test of Redox can be performed using Methylene Blue and any reducer such as powdered Wonder Shell. Simply add enough Methylene Blue 8 oz. of water to turn the water a brilliant blue (usually one drop). Then add about ½ teaspoon powdered Wonder Shell to the 8 oz. of water and stir. The Methylene Blue will dissipate due to the reducer.
Taking this test a step further into the real world; again, add enough Methylene Blue 8 oz. of water to turn the water a brilliant blue (again one drop), this will be the control color. Then take out 8 oz. of aquarium water and add the same amount of Methylene Blue as in the control glass of water. If the color dissipates at all (even the slightest) you probably have a balanced Redox with at least some reduction. Expect to see little color change in a healthy aquarium, although absolutely no color change MAY indicate too much oxidation, while any more than a subtle change may indicate too much reduction.
More about Methylene Blue:
• Aquarium Medications Part 3, Methylene Blue
For a more scientific Redox measurement device:
• Professional Waterproof Portable pH/ORP Meter
The picture above graphically displays the dramatic difference. In the control tank (left) and tank with a level one capable UVC Sterilizer (AAP 7-Watt Compact UV), potassium permanganate (which is a strong oxidizer) was added at the same rate. Potassium Permanganate reduces from a red color to brown to clear. It is very obvious the difference in the two tanks, which means that there is less oxidative stress to fish. Thus, this increases the fish' ability fight off diseases in particular diseases such as Lateral Line disease in marine aquariums which seem to be more prevalent when oxidative stress is higher.
While there is some criticism of UV Sterilizer use claiming if used to the point of killing or altering all potential pathogens, this would be detrimental similar to never exposing your children to anything and keeping them isolated in a bubble. This is only correct in that the immune system should not be sheltered from every possibly malady so as to be able to provide a healthy immune response when needed.
However, the complete destruction of all pathogens is not what happens with correct UV Sterilizer use.
First this is not possible for even the best UV to destroy all pathogens in an aquarium or pond (or altering pathogens making them unable to multiply) as for one not all pathogens are in the water column, many or already opportunistic living in the fish, substrate, or elsewhere.
What a well-designed and properly installed true UV Sterilizer can do is improve immune function via improved Redox Balance. This aspect of a UV Sterilizer is so often missed or even utilized by those who own a true UV Sterilizer but only run a UV after a problem has occurred. This is similar to how one might give their children Echinacea before sending them off to school since Echinacea improves immune response, but in itself generally does not destroy disease pathogens.
However, it is noteworthy that the use of Ozone Generators does NOT accomplish the same results with Redox balance, despite similar results in sterilization (I found this stated in some very confused articles and forum posts I have read).
Product Resource:
• Aquarium/Pond UV Sterilizers CAPABLE of improving Redox
Lighting and the "quality" of this light also impact aquarium Redox. By quality I mean the nanometer wavelengths make a difference on the impact of Redox, with lighting in a more natural "noon time sun" 6500K appearing to have the greatest impact overall. This impact is a reduction of ORP from the time the lights go on in the morning into the day, going back up after lights are off.
The way the redox value changes with light is due to direct molecular bond changes from photon impact. The result of this is an atom/molecule with a higher or lower oxidation state, which is then shown as a change in measured redox value. It's a small but important distinction. Redox is a chemical reaction, while irradiation is direct manipulation of the bonds without a chemical reaction.
The implication is that if you need a higher ORP for oxidation due to organic waste buildup, lights should be reduced. However generally speaking and if your fish are sick, reducing Redox (ORP) is what you want and stronger and better lighting (such as produced by the AquaRay GroBeams and AquaBeams).
Recently reviewed and updated tests have also shown the positive benefit of near infrared to infrared light in the improvement of Redox Reduction and wound healing. Again, this is now a proven fact that has aquatic implications for good lighting that hits these nanometer spikes. Generally, within 5500K to 6700K lighting, although specific 'Red' lighting can help too, such as the TMC Flexi-Red LEDs or TMC HO Colour Plus LEDs.
Product Resources:
• TMC Aquarium LED Lights; Including Colour Plus
See:
• Wise and limited use of Ozoneizers/Ozone Generators (Red Light Therapy)
• Aquarium Lighting; Facts & Information
Important: What is often missed, is that although you may test and, for instance find, high calcium levels in an aquarium via a GH over 300, this does not necessarily mean you have any Redox reduction as I should point out that very basic fact of Redox reduction is the donor (such as calcium) transfers electrons to another substance, and is thus, oxidized itself. This is VERY key, meaning your GH test (or Calcium, Magnesium, etc tests in saltwater) may tell you that you have ample or even too much calcium. HOWEVER, these calcium molecules may have already given up their electrons to other molecules such as fish cell molecules so as to achieve a state of balance in aquarium Redox.
This again is where many will claim that their calcium or other minerals are high enough or even too high (as many still get INCORRECTLY stressed about the high GH brought on by AAP Wonder Shells, totally missing that these mineral blocks are constantly reducing the water via their donation of electrons!).
This is also where water changes and UV Sterilization also help as these processes constantly balance out Redox. Since water changes (which also aid in oxidation, which is the other side of the equation) can only be done in intervals. Unless you have a flow through system, the use of mineral blocks, or other water conditioners as well as UV Sterilization to keep a steadier flow of electrolyte donation is important.
With the above statements in mind, one must look at two VERY basic organic chemical reactions that occur in an aquarium: As stated in the opening section of this article; “oxidation of carbon to yield carbon dioxide”, this describes the process of nitrification or in other words the breakdown of organic wastes by Autotrophic nitrifying bacteria into ammonia and nitrites.
The higher the bio load, the more of this process is occurring. So, it is imperative to replace elements such as negatively charged ions of calcium, magnesium, etc. that are oxidized due to this process.
This is where many aquarists “miss the boat” as per reasons for water changes, replenishing minerals, or other Redox Reduction processes that will balance this out. This is also why tanks without good, ionized mineralization, UV Sterilization, water changes, etc. will often be MUCH less healthy as per disease resistance.
Reduction as also stated in the opening section: “reduction of carbon by hydrogen to yield methane (CH4)”, this describes the de-nitrification of nitrate as it reduces to nitrogen. This process is more common in Marine tanks but can take place in freshwater in certain places where methane production is not an issue such as filters with anaerobic filtration that allow methane to escape (volcanic rock & SeaChem Matrix provide a good place for this process). Also, the growth of plants, especially with strong roots in a sandy substrate allows for de-nitrification/Redox reduction. This is an area where Veggie Filters in Ponds shine in their ability to maintain excellent bio parameters. This also to a lesser degree shows where some green algae growth is not all that bad.
Further Reference:
• Pond Veggie/Bog Filters
All this said, Reduction generally de-nitrification processes CANNOT keep up with oxidation processes necessary for breakdown of organic wastes, which is why help is needed via water changes, mineralization, UV Sterilization, etc. This is where many naysayers of products such as Wonder Shells, or UV Sterilizers TOTALLY miss the point as to how much they can improve water quality. This is also how the use of products such as Volcanic Rock or SeaChem Matrix can also indirectly affect Redox.
Product Resources:
• Volcanic Rock Filter Media, Nitrifying, De-nitrifying
• SeaChem Matrix; Biological Nitrate Remover, De-nitrifier
Important! Put another way, there is certainly a time and place for a high Redox (oxidation) as lower oxygen levels from many causes such as high organic decomposition is generally not desirable, and this will lower your Redox oxidation side of the equation. This is not what I recommend as a means of a reducing Redox environment. However, to not have the proper reducers or creating a highly oxidative environment is also not good for long term fish health.
The Redox Potential is always going to be in flux and as, yet no scientific equilibrium has been established here. Maintaining reducers such as Calcium and Magnesium in your aquarium is just as important as proper levels of dissolved oxygen (which raises Redox). There is evidence both anecdotal and controlled that proper levels of calcium and other antioxidants such as Vitamin C (along with other healthy water parameters) will prevent and even cure Hole in the Head disease (HITH). Redox is an important parameter here.
Further Reference:
• Aquarium Answers, about Hole in the Head Disease
As stated earlier, Redox can be more important than pH, there are definitely correlations to pH and the Redox effect on pH. But you cannot make the assumption that if you have a pH of 8.0, that you have good reduction. Just like with GH, where your GH is high, you can still have low pH. The bottom line is that there are relations between Redox, GH, pH, and even kH, but these are still individual water parameters.
Also as noted in the beginning of this article, this is not a parameter to stress over, HOWEVER, Redox reduction should NOT be blown off as many anecdotal/uninformed commentators do in some aquatic forums. The more I have studied and observed in this subject, the more I feel it should not be ignored!
Effect on Fish: Active oxygen molecules, or free radicals, are produced in the water and the fish’ body. They are extremely reactive and can also attach themselves to normal, healthy cells and damage them genetically. These active oxygen radicals steal electrons from normal, healthy biological molecules.
This electron theft by active oxygen oxidizes tissue and can cause disease; these are proven facts!
This why the constant hammering by some un-informed aquarists (such a popular planted aquarium guru) against UVs, too high a GH (which is not always a good indicator of electrolyte balance anyway), and even the use of mineral blocks (such as Wonder Shells) is ludicrous! Your GH may test higher than desired but in actuality is maintaining a healthy Reducing Redox via the reducing properties of positively charged calcium, magnesium, etc.
Human studies show a clear link between the Redox level in the blood of a healthy person and an unhealthy person, especially when it comes to cancer. Those with cancer and other chronic illnesses show more oxidation. For fish, this means that while oxidizing agents have their place, we should not be regularly dumping oxidizing products in our aquariums, even if they are so-called "natural" products whether it be Microbe-Lift Herbtana or API Melafix. The scientific facts are that over the long term the regular use of these products WILL SHORTEN the life of your fish and make them more susceptible to diseases, including tumors.
Reference:
• Tissue redox activity as a hallmark of carcinogenesis
In short, oxidative stress most definitely plays a role in both short- & long-term fish health and ability to heal. Diseases such as Columnaris and HLLE can be linked to oxidative stress from both my 1000s of observations as well as research.
Effect on plants: In a controlled test conducted with terrestrial plants; plants treated with magnetized water (basically Redox Balanced Water) reacted in a test, with a growth in the order of 20% to 40% bigger than the plants treated with regular water. While no test has been conducted with planted aquariums, it is not rocket science to draw conclusions from this study and apply this to a planted freshwater aquarium and the effect of Redox Balance on plant growth!
Simple Redox Test: A simple test of Redox can be performed using Methylene Blue and any reducer such as powdered Wonder Shell. Simply add enough Methylene Blue 8 oz. of water to turn the water a brilliant blue (usually one drop). Then add about ½ teaspoon powdered Wonder Shell to the 8 oz. of water and stir. The Methylene Blue will dissipate due to the reducer.
Taking this test a step further into the real world; again, add enough Methylene Blue 8 oz. of water to turn the water a brilliant blue (again one drop), this will be the control color. Then take out 8 oz. of aquarium water and add the same amount of Methylene Blue as in the control glass of water. If the color dissipates at all (even the slightest) you probably have a balanced Redox with at least some reduction. Expect to see little color change in a healthy aquarium, although absolutely no color change MAY indicate too much oxidation, while any more than a subtle change may indicate too much reduction.
More about Methylene Blue:
• Aquarium Medications Part 3, Methylene Blue
For a more scientific Redox measurement device:
• Professional Waterproof Portable pH/ORP Meter
Effect of Stress
In biological systems, the Redox Potential must be kept reduced to perpetuate the life process. In other words, the concentration of DHA (an oxidized form of ascorbic acid), must be kept to a minimum. During injuries or stress which expose body tissues to atmospheric oxygen or disease, several reactions are triggered in polyunsaturated fatty acids (PUFAs) present in cellular membranes which produce hormones called prostaglandins that repair damage. Under oxidizing stress (injury or disease) or impaired ability to manufacture NADH (aging or disease), the concentration of DHA can rise as the redox potential slips. This is a potentially life-threatening state for fish or any other animal.
The initial rate at which the redox potential falls as DHA increase is initially slow, however as DHA increases further, the redox potential falls faster, becoming progressively more antagonistic to the metabolic processes of life. Eventually; if the process is not stopped, the fish (or any animal organism) slides down into death.
This is why even prolonged stress can lead to illness and even death and why under stressful conditions having ample reducers such as a constant supply of positively charged calcium ions or TRUE UVC sterilization is so important! Common stressor examples include constant harassment by tank mates, or constant, albeit well meaning use of many treatments meant to cure fish, but that are also Redox Oxidizers.
From Wikipedia: "Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species and a biological system's ability to readily detoxify the reactive intermediates or to repair the resulting damage. Disturbances in the normal redox state of cells can cause toxic effects through the production of peroxides and free radicals that damage all components of the cell, including proteins, lipids, and DNA. Further, some reactive oxidative species act as cellular messengers in redox signaling. Thus, oxidative stress can cause disruptions in normal mechanisms of cellular signaling."
Reference Source:
• Wikipedia; Oxidative stress
The initial rate at which the redox potential falls as DHA increase is initially slow, however as DHA increases further, the redox potential falls faster, becoming progressively more antagonistic to the metabolic processes of life. Eventually; if the process is not stopped, the fish (or any animal organism) slides down into death.
This is why even prolonged stress can lead to illness and even death and why under stressful conditions having ample reducers such as a constant supply of positively charged calcium ions or TRUE UVC sterilization is so important! Common stressor examples include constant harassment by tank mates, or constant, albeit well meaning use of many treatments meant to cure fish, but that are also Redox Oxidizers.
From Wikipedia: "Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species and a biological system's ability to readily detoxify the reactive intermediates or to repair the resulting damage. Disturbances in the normal redox state of cells can cause toxic effects through the production of peroxides and free radicals that damage all components of the cell, including proteins, lipids, and DNA. Further, some reactive oxidative species act as cellular messengers in redox signaling. Thus, oxidative stress can cause disruptions in normal mechanisms of cellular signaling."
Reference Source:
• Wikipedia; Oxidative stress
Healthy Redox Methods
Proper water changes with ion balanced clean water (electrolytes) with a GH of at least 100- 200 (GH will supply many important reducers such as Calcium & Magnesium) and with a kH of at least 50-100 ppm (for control of nitric acid production).
A proper Reducing/Balanced Redox is another reason for water changes and is also a major reason to not use drinking water intended for human consumption, softened water, or RO/DI water unless it is properly re-mineralized. Often even well or tap water is not ionically balanced. Use of un-mineralized water or water missing mineral Cations will have no positive affect on Redox. It is also noteworthy that my tests many years back indicated that water changes by themselves do NOT solve the Redox balance equation unless very large 50% water changes or higher with ionically balanced water are done weekly or even more often, and even then, adding oxidizing additives in between can even nullify these large water changes!
Removal of organic mulm during cleanings is important as this leads to nitric acid production and high DOC (dissolved organic compounds) which are major contributors to a poor Redox Balance. If your water changes are not effective in removing all organic mulm, consider a re-circulating vacuum (such as an Eheim Sludge Remover) that can drain and then continue to remove mulm that is easily emptied from the micron filter.
Product Resource:
• Eheim Sludge Remover; Aquarium Organic Debris Removal Tool
Further Resources:
• Aquarium Cleaning, Water Changes
• Aquarium Answers, Fish Drinking, Osmoregulation
Also ponds with “flow through” or spring fed water sources will maintain a good Redox Potential by virtue of the constant addition of fresh, clean and mineralized water, which is why many lakes will become less “healthy” during the late summer months when stream that feed them “dry up”.
Good aquarium circulation This aids both sides of the Redox equation, although more so the oxidation side.
A reasonable supply of dissolved oxygen provided by diffusers on power heads, internal filter, air stones, spray bars on the returns of canister filters, etc. Just make sure you are doing a good job of breaking the surface tension of the water, as this is where gasses, including oxygen are exchanged!
Adequate Calcium, Magnesium, sodium, and general electrolyte levels, THIS IS VERY IMPORTANT! As noted earlier, this is an on-going process with a starting point to maintain a GH of 100 to 500 ppm (less for soft water fish, higher yet for African Cichlids and Marine). This is an incomplete picture as recent evidence shows that you may have an adequate amount of Calcium as per your test, but this calcium has given up many of the available electrons and is not aiding in your Redox balance. This is why using mineral blocks or similar concepts is so important. This also further disproves the point of many old aquarium myths about too high of a GH, as what your test is measuring may not be the whole picture since it cannot measure positive mineral ions that may or may not be present.
AAP Wonder Shells are very useful since they add important mineral cations on a constant basis. As well, products such as SeaChem Replenish, Instant Amazon, or Aragamix can be mixed with water and dripped into aquariums to achieve constant positive mineral ions. However, I have not found that the use of crushed coral will dissolve at a rate to supply enough of these minerals to have much effect on Redox Balance. As noted earlier in this article, a test before and after using an aquarium water sampling with AAP Wonder Shell dust/shaving, showed an immediate +20% drop in Redox (ORP)
Product Resources:
• Unique/Fresh AAP Wonder Shells
• SeaChem Replenish
Another potential source is an ionic plenum filter, utilizing Calcium Bentonite (Cat Litter often contains this). Calcium Bentonite is a natural clay formed from the weathering of volcanic ash in the presence of water and has the potential of adding essential mineral ions back into the water column.
I have a good article dealing with GH, Calcium, and Magnesium here:
• CALCIUM, KH, AND MAGNESIUM IN AQUARIUMS; The importance of mineral ions
A UV Sterilizer is one of the more predictable and proven tools here (when CORRECTLY installed with a Level One UV Sterilizer), but also not a cure all to otherwise poor conditions. UVs work two ways in my research; [A] they morph oxygen (O2) molecules into Ozone (O3), the O3 quickly degrades, the UV then [B] transfers electrons to substances, thus reducing.
As a side note it is the Ozone layer that blocks many harmful UV rays from reaching the Earth, this is due to the reaction of UVC and Ozone high in our atmosphere, so by using a UV Sterilizer (which has safely contained UVC irradiation), you break down radicals such as Ozone in your aquarium.
As noted earlier in this article, please reference this article for more about this important subject:
• Aquarium & Pond UV Sterilization
Wise and limited use of most aquarium treatments. As has been noted earlier in this article, many treatments/medications are in fact oxidizers and in fact effect a cure by being less of an oxidizing stressor on the fish/patient than the pathogen, sore, infection, etc. Common treatments that are oxidizers include malachite green, potassium permanganate, coper sulfate and even methylene blue. This is not limited to chemical treatments either, in fact just because something is natural/organic does not mean it cannot be an oxidizer and cause oxidative stress. This includes products such as Pimafix, Melafix, Herbtana, Artemiss and much more.
This does not mean any of the above-mentioned products (which is far from a complete list) should never be used, just these should not be used under the mistaken presumption that these will help with fish stress (quite the opposite based on the black and white facts of chemistry), nor that having these constantly present in the aquarium is good for the fish: IT IS NOT! Unfortunately, many discounters selling Herbtana recommend this product to support the immune system, which IT DOES NOT!
Medicated Wonder Shells are a product that buffers the oxidizing affect of of the medications present within the calcium carbonate, but even these should never be in constant use, but better these than constantly adding many other chemical and so-called natural products that are in fact oxidizers!
Wise and limited use of Ozonizers/Ozone Generators. While popular among some in aquarium keeping, especially in large scale operations, care should be taken when using these devices as they do in fact still affect a healthy aquarium Redox. My observations, as well those of others confirm that when Ozonizers/Ozone Generators are used, symptoms of oxidative stress increase, including HLLE disease in marine fish.
Further Reading:
• Lateral Line Disease in Fish (HLLE)
Best use practices are along with a well-designed Protein Skimmer so as to improve its performance where the effect on the aquarium water column is minimized. Use of a Ozonizer/Ozone Generator for germicidal reasons is in my experience best left to a well-designed high dwell time UV Sterilizer.
Good Lighting: Although not as common a problem for most marine aquarists, especially reef keepers as these aquarists usually have (& know the benefits) of good lighting, in freshwater aquariums this is often a contributing factor to a poor Redox (and to other problems as well).
The reasons are admittedly not completely clear, however from my observations, tests and research, I believe correct lighting plays a role similar to UV Sterilization as explained above, when lighting with poor PAR qualities which are generally found in lights with approximately 6500 Kelvin temperatures. My suspicions are that although PAR has been scientifically established to be important for plants, it may also play an important role for fish as well (both in electromagnetism of the water and similar to how humans derive the benefit of vitamin D production from sunlight).
As noted earlier, the red spectrum has also been recently proven to aid in Redox Reduction and healing of wounds, so this is another reason to maintain good lighting, not just for your reef inhabitants or planted freshwater aquariums but for your guppies, goldfish, etc.!!!
It is also noteworthy that in tests using a Redox meter, we found that full spectrum aquarium lighting lowered Redox, acting like a reducer (similar to how a UV Sterilizer acts on Redox).
Reference:
• Aquarium Lighting Facts & Information
See Also:
• Wise and Limited Use of Ozonizers/Ozone Generators (Red Light Therapy)
Use of water conditioners (for use in chlorine/chloramine removal) during times of stress (especially oxidative stress) such as SeaChem Prime which are mostly all Reducers. However, these products only temporarily reduce Redox and are not a long-term solution. It also should be noted that aquarium water conditioners will often cause temporary cloudiness of aquariums due to their strong reducing abilities, however this cloudiness is short lived in healthy aquariums.
Product Resource:
• SeaChem Prime; Water Conditioner, Reducer
Another danger is the use of Vitamin C as a water conditioner for large percentage water changes (in particular as ascorbic acid); while this is very effective for immediate chlorine removal, it will also drastically drop pH a point or more which can add considerably to stress.
For more about aquarium water conditioners, please read these articles:
• Tap water in Aquarium/Pond; Vitamin C Use
• Aquarium Water Conditioners
Use of Fish Food with Optimized Energy Levels: While well established in human nutrition, many are still not aware of the facts around free radicals and how the breakdown of fish food for energy contributes to this in fish as well, despite research by ufl.edu and ongoing research by fish food guru Clay Neighbors.
"Free radicals are highly unstable molecules that are naturally formed when the fish converts food into energy. Free radicals can cause 'oxidative stress,' a process that can trigger cell damage, therefore limiting these free radicals by an optimized energy level will benefit your fish long term".
Product Resource:
• Clay Neighbor's Energy Level Optimized Fish Food
Reference, further reading:
• Fish Nutrition
A proper Reducing/Balanced Redox is another reason for water changes and is also a major reason to not use drinking water intended for human consumption, softened water, or RO/DI water unless it is properly re-mineralized. Often even well or tap water is not ionically balanced. Use of un-mineralized water or water missing mineral Cations will have no positive affect on Redox. It is also noteworthy that my tests many years back indicated that water changes by themselves do NOT solve the Redox balance equation unless very large 50% water changes or higher with ionically balanced water are done weekly or even more often, and even then, adding oxidizing additives in between can even nullify these large water changes!
Removal of organic mulm during cleanings is important as this leads to nitric acid production and high DOC (dissolved organic compounds) which are major contributors to a poor Redox Balance. If your water changes are not effective in removing all organic mulm, consider a re-circulating vacuum (such as an Eheim Sludge Remover) that can drain and then continue to remove mulm that is easily emptied from the micron filter.
Product Resource:
• Eheim Sludge Remover; Aquarium Organic Debris Removal Tool
Further Resources:
• Aquarium Cleaning, Water Changes
• Aquarium Answers, Fish Drinking, Osmoregulation
Also ponds with “flow through” or spring fed water sources will maintain a good Redox Potential by virtue of the constant addition of fresh, clean and mineralized water, which is why many lakes will become less “healthy” during the late summer months when stream that feed them “dry up”.
Good aquarium circulation This aids both sides of the Redox equation, although more so the oxidation side.
A reasonable supply of dissolved oxygen provided by diffusers on power heads, internal filter, air stones, spray bars on the returns of canister filters, etc. Just make sure you are doing a good job of breaking the surface tension of the water, as this is where gasses, including oxygen are exchanged!
Adequate Calcium, Magnesium, sodium, and general electrolyte levels, THIS IS VERY IMPORTANT! As noted earlier, this is an on-going process with a starting point to maintain a GH of 100 to 500 ppm (less for soft water fish, higher yet for African Cichlids and Marine). This is an incomplete picture as recent evidence shows that you may have an adequate amount of Calcium as per your test, but this calcium has given up many of the available electrons and is not aiding in your Redox balance. This is why using mineral blocks or similar concepts is so important. This also further disproves the point of many old aquarium myths about too high of a GH, as what your test is measuring may not be the whole picture since it cannot measure positive mineral ions that may or may not be present.
AAP Wonder Shells are very useful since they add important mineral cations on a constant basis. As well, products such as SeaChem Replenish, Instant Amazon, or Aragamix can be mixed with water and dripped into aquariums to achieve constant positive mineral ions. However, I have not found that the use of crushed coral will dissolve at a rate to supply enough of these minerals to have much effect on Redox Balance. As noted earlier in this article, a test before and after using an aquarium water sampling with AAP Wonder Shell dust/shaving, showed an immediate +20% drop in Redox (ORP)
Product Resources:
• Unique/Fresh AAP Wonder Shells
• SeaChem Replenish
Another potential source is an ionic plenum filter, utilizing Calcium Bentonite (Cat Litter often contains this). Calcium Bentonite is a natural clay formed from the weathering of volcanic ash in the presence of water and has the potential of adding essential mineral ions back into the water column.
I have a good article dealing with GH, Calcium, and Magnesium here:
• CALCIUM, KH, AND MAGNESIUM IN AQUARIUMS; The importance of mineral ions
A UV Sterilizer is one of the more predictable and proven tools here (when CORRECTLY installed with a Level One UV Sterilizer), but also not a cure all to otherwise poor conditions. UVs work two ways in my research; [A] they morph oxygen (O2) molecules into Ozone (O3), the O3 quickly degrades, the UV then [B] transfers electrons to substances, thus reducing.
As a side note it is the Ozone layer that blocks many harmful UV rays from reaching the Earth, this is due to the reaction of UVC and Ozone high in our atmosphere, so by using a UV Sterilizer (which has safely contained UVC irradiation), you break down radicals such as Ozone in your aquarium.
As noted earlier in this article, please reference this article for more about this important subject:
• Aquarium & Pond UV Sterilization
Wise and limited use of most aquarium treatments. As has been noted earlier in this article, many treatments/medications are in fact oxidizers and in fact effect a cure by being less of an oxidizing stressor on the fish/patient than the pathogen, sore, infection, etc. Common treatments that are oxidizers include malachite green, potassium permanganate, coper sulfate and even methylene blue. This is not limited to chemical treatments either, in fact just because something is natural/organic does not mean it cannot be an oxidizer and cause oxidative stress. This includes products such as Pimafix, Melafix, Herbtana, Artemiss and much more.
This does not mean any of the above-mentioned products (which is far from a complete list) should never be used, just these should not be used under the mistaken presumption that these will help with fish stress (quite the opposite based on the black and white facts of chemistry), nor that having these constantly present in the aquarium is good for the fish: IT IS NOT! Unfortunately, many discounters selling Herbtana recommend this product to support the immune system, which IT DOES NOT!
Medicated Wonder Shells are a product that buffers the oxidizing affect of of the medications present within the calcium carbonate, but even these should never be in constant use, but better these than constantly adding many other chemical and so-called natural products that are in fact oxidizers!
Wise and limited use of Ozonizers/Ozone Generators. While popular among some in aquarium keeping, especially in large scale operations, care should be taken when using these devices as they do in fact still affect a healthy aquarium Redox. My observations, as well those of others confirm that when Ozonizers/Ozone Generators are used, symptoms of oxidative stress increase, including HLLE disease in marine fish.
Further Reading:
• Lateral Line Disease in Fish (HLLE)
Best use practices are along with a well-designed Protein Skimmer so as to improve its performance where the effect on the aquarium water column is minimized. Use of a Ozonizer/Ozone Generator for germicidal reasons is in my experience best left to a well-designed high dwell time UV Sterilizer.
Good Lighting: Although not as common a problem for most marine aquarists, especially reef keepers as these aquarists usually have (& know the benefits) of good lighting, in freshwater aquariums this is often a contributing factor to a poor Redox (and to other problems as well).
The reasons are admittedly not completely clear, however from my observations, tests and research, I believe correct lighting plays a role similar to UV Sterilization as explained above, when lighting with poor PAR qualities which are generally found in lights with approximately 6500 Kelvin temperatures. My suspicions are that although PAR has been scientifically established to be important for plants, it may also play an important role for fish as well (both in electromagnetism of the water and similar to how humans derive the benefit of vitamin D production from sunlight).
As noted earlier, the red spectrum has also been recently proven to aid in Redox Reduction and healing of wounds, so this is another reason to maintain good lighting, not just for your reef inhabitants or planted freshwater aquariums but for your guppies, goldfish, etc.!!!
It is also noteworthy that in tests using a Redox meter, we found that full spectrum aquarium lighting lowered Redox, acting like a reducer (similar to how a UV Sterilizer acts on Redox).
Reference:
• Aquarium Lighting Facts & Information
See Also:
• Wise and Limited Use of Ozonizers/Ozone Generators (Red Light Therapy)
Use of water conditioners (for use in chlorine/chloramine removal) during times of stress (especially oxidative stress) such as SeaChem Prime which are mostly all Reducers. However, these products only temporarily reduce Redox and are not a long-term solution. It also should be noted that aquarium water conditioners will often cause temporary cloudiness of aquariums due to their strong reducing abilities, however this cloudiness is short lived in healthy aquariums.
Product Resource:
• SeaChem Prime; Water Conditioner, Reducer
Another danger is the use of Vitamin C as a water conditioner for large percentage water changes (in particular as ascorbic acid); while this is very effective for immediate chlorine removal, it will also drastically drop pH a point or more which can add considerably to stress.
For more about aquarium water conditioners, please read these articles:
• Tap water in Aquarium/Pond; Vitamin C Use
• Aquarium Water Conditioners
Use of Fish Food with Optimized Energy Levels: While well established in human nutrition, many are still not aware of the facts around free radicals and how the breakdown of fish food for energy contributes to this in fish as well, despite research by ufl.edu and ongoing research by fish food guru Clay Neighbors.
"Free radicals are highly unstable molecules that are naturally formed when the fish converts food into energy. Free radicals can cause 'oxidative stress,' a process that can trigger cell damage, therefore limiting these free radicals by an optimized energy level will benefit your fish long term".
Product Resource:
• Clay Neighbor's Energy Level Optimized Fish Food
Reference, further reading:
• Fish Nutrition
Redox Problems
Driftwood: Make sure you soak all Mango Wood or Driftwood for several days in a solution of 2 tablespoons pure salt to 2-5 gallons of water to leach out impurities. Even then, if you continue to have problems remove the wood. Overuse of peat can also cause similar problems.
CO2 generators if not kept at a balance can also cause problems with the Redox Reduction abilities, this is rare, but worth considering.
A good link to help here is:
• Measuring CO2 levels in a Planted Tank
An abundance of organic mulm in filters or substrate. This can be wet/dry filters, canister filters, and very often in a poorly managed "live rock reef" and sand in a reef aquarium. I will also note as per my many tests with goldfish aquariums, I found Under Gravel Filters the worst offenders here with high production of DOC (Dissolved organic compounds), and even with UV Sterilizers these tanks were more difficult to maintain a balanced Redox environment in. Check your cleaning procedures as well.
This mulm or DOC has a direct impact on your aquarium bio load, resulting in a fragile Redox Balance whereby any change to this balance, often a sudden increase in Redox Reduction by the addition of a properly installed UV Sterilizer results in noticeable change to water quality.
Please Reference:
• Aquarium Bio Load
A lack of electrolytes (positively charged mineral ions), as mentioned earlier, 'AAP Wonder Shells' or dripping products such as Aragamix, SeaChem Replenish, or similar can help here, BUT they are not a cure all for other problems.
A tank with high nitrates (nitrates are a mild oxidant) will also tend to be high in nitric acid (but not always); a properly cycled tank is important! This also usually occurs with high DOC levels (dissolved organic compounds), regular water changes that remove organic mulm are important here.
Overuse of many natural remedies. While many think that natural remedies have no side effects, they are incorrect, at least when it comes to Redox. Some extensively tested examples include Melafix, Pimafix, with newer testing revealing more such as Herbtana.
I am not saying these are not viable alternatives to conventional treatments, but many use organics that go after bacteria and parasites in ways that alter pH and more importantly Redox as they are oxidizers. The problem occurs with the constant use of these products thinking they are 100% safe when they are not. No treatment is 100% safe and has some sort of side effect.
As an example, a Medicated Wonder Shell actually improves Redox, but some of the chemicals contained in these can be harmful to plants if used constantly. The bottom line is to use conservatively and only when necessary!!!
CO2 generators if not kept at a balance can also cause problems with the Redox Reduction abilities, this is rare, but worth considering.
A good link to help here is:
• Measuring CO2 levels in a Planted Tank
An abundance of organic mulm in filters or substrate. This can be wet/dry filters, canister filters, and very often in a poorly managed "live rock reef" and sand in a reef aquarium. I will also note as per my many tests with goldfish aquariums, I found Under Gravel Filters the worst offenders here with high production of DOC (Dissolved organic compounds), and even with UV Sterilizers these tanks were more difficult to maintain a balanced Redox environment in. Check your cleaning procedures as well.
This mulm or DOC has a direct impact on your aquarium bio load, resulting in a fragile Redox Balance whereby any change to this balance, often a sudden increase in Redox Reduction by the addition of a properly installed UV Sterilizer results in noticeable change to water quality.
Please Reference:
• Aquarium Bio Load
A lack of electrolytes (positively charged mineral ions), as mentioned earlier, 'AAP Wonder Shells' or dripping products such as Aragamix, SeaChem Replenish, or similar can help here, BUT they are not a cure all for other problems.
A tank with high nitrates (nitrates are a mild oxidant) will also tend to be high in nitric acid (but not always); a properly cycled tank is important! This also usually occurs with high DOC levels (dissolved organic compounds), regular water changes that remove organic mulm are important here.
Overuse of many natural remedies. While many think that natural remedies have no side effects, they are incorrect, at least when it comes to Redox. Some extensively tested examples include Melafix, Pimafix, with newer testing revealing more such as Herbtana.
I am not saying these are not viable alternatives to conventional treatments, but many use organics that go after bacteria and parasites in ways that alter pH and more importantly Redox as they are oxidizers. The problem occurs with the constant use of these products thinking they are 100% safe when they are not. No treatment is 100% safe and has some sort of side effect.
As an example, a Medicated Wonder Shell actually improves Redox, but some of the chemicals contained in these can be harmful to plants if used constantly. The bottom line is to use conservatively and only when necessary!!!
Overuse of activated carbon and especially products such as Chemipure, Poly Pads, & Purigen.
While these products have their place, and in aquariums such as marine aquariums where there are likely more mineral Cations available to balance their use, these products generally do not present much of a problem with reasonable use. However, with copious amounts of activated carbon, or even low use of products such as Chemipure especially in aquariums already conducive to poor Redox balance such as low pH and high organic load Amazon River biotope aquariums, these can have a notable impact on Redox Balance and fish health!
Improper use of an Ozonizer (Ozone Generator) can defeat the purpose of using a UV Sterilizer by allowing ozone into an aquarium and thus raising the Redox to levels too high for long term fish health.
An Ozone Generator MUST be used with a Protein Skimmer that is capable of containing ALL ozone, such as the TMC V2 Skim Protein Skimmer. This is not the case with many Protein Skimmers, and even the TMC model or others similar must be properly monitored when using ozone.
Be careful with overuse of CO2. Generally, CO2 use is not a problem. However, if your aquarium is already struggling with Redox balance including the lack of constant mineral Cations, the use of Carbon Dioxide can lead to increased oxidative stress. During photosynthesis, the oxidized P680 (P680+) is subsequently reduced by an electron originating from water, which if your aquarium redox lacks sufficient reducers to give up this electron, can result in oxidative stress (the use of AAP Wonder shells can help here).
The facts are that CO2 can also create carbonic acid which will act as an oxidizing agent. As well Carbon dioxide can directly act as an oxidizing agent. You can see it clearly acting like one in the following reaction, where it oxidizes magnesium to form MgO(s):
• Mg(s) + 12
• CO2 ? MgO(s) + 12C(s)
References:
• Magnesium Burns in Carbon Dioxide (reaction only)
• P680, or Photosystem II primary donor
As noted earlier, good lighting and the addition of a UV Sterilizer can play an important role for a balanced aquatic Redox Potential as newer studies have proven that UV Sterilization increases immunity by adding electrons and balancing the essential electromagnetism of the water. Although less conclusive at this time, high PAR lighting (usually found in lights with an approximate Kelvin of 6500 K), also aids in replenishing reducing electrons due to depleted reducing minerals.
Product Resource:
• True UV Sterilizers Capable of improving Redox Balance
While these products have their place, and in aquariums such as marine aquariums where there are likely more mineral Cations available to balance their use, these products generally do not present much of a problem with reasonable use. However, with copious amounts of activated carbon, or even low use of products such as Chemipure especially in aquariums already conducive to poor Redox balance such as low pH and high organic load Amazon River biotope aquariums, these can have a notable impact on Redox Balance and fish health!
Improper use of an Ozonizer (Ozone Generator) can defeat the purpose of using a UV Sterilizer by allowing ozone into an aquarium and thus raising the Redox to levels too high for long term fish health.
An Ozone Generator MUST be used with a Protein Skimmer that is capable of containing ALL ozone, such as the TMC V2 Skim Protein Skimmer. This is not the case with many Protein Skimmers, and even the TMC model or others similar must be properly monitored when using ozone.
Be careful with overuse of CO2. Generally, CO2 use is not a problem. However, if your aquarium is already struggling with Redox balance including the lack of constant mineral Cations, the use of Carbon Dioxide can lead to increased oxidative stress. During photosynthesis, the oxidized P680 (P680+) is subsequently reduced by an electron originating from water, which if your aquarium redox lacks sufficient reducers to give up this electron, can result in oxidative stress (the use of AAP Wonder shells can help here).
The facts are that CO2 can also create carbonic acid which will act as an oxidizing agent. As well Carbon dioxide can directly act as an oxidizing agent. You can see it clearly acting like one in the following reaction, where it oxidizes magnesium to form MgO(s):
• Mg(s) + 12
• CO2 ? MgO(s) + 12C(s)
References:
• Magnesium Burns in Carbon Dioxide (reaction only)
• P680, or Photosystem II primary donor
As noted earlier, good lighting and the addition of a UV Sterilizer can play an important role for a balanced aquatic Redox Potential as newer studies have proven that UV Sterilization increases immunity by adding electrons and balancing the essential electromagnetism of the water. Although less conclusive at this time, high PAR lighting (usually found in lights with an approximate Kelvin of 6500 K), also aids in replenishing reducing electrons due to depleted reducing minerals.
Product Resource:
• True UV Sterilizers Capable of improving Redox Balance
Summary
This was in the past a relatively unexplored area of fish keeping that has many implications including prevention of HLLE in marine fish. As well there is still a lot to be learned here and I have to admit having to change my opinions based on newer evidence (I too thought that ONLY a high positive Redox/ORP should be maintained), my research is still ongoing and I try and read whatever I can on this subject, especially from outside the aquarium hobby, even if it may not fit with theories I hold.
In fact, this is now probably my most researched subject, with many outside sources cited here. It is also noteworthy that many concepts applied here to explain my researched results are readily explained in what is now common knowledge as per oxidative stress in humans (this includes diet). As well, I have received emails from outside the aquarium community from medical researchers congratulating my conclusions as to Redox by citing current research and then applying it to aquariums.
Unfortunately, some aquarists in their writings or anecdotal forum comments fail to look at the research that has gone on the subject of Redox and stick to their old tired theories about Aquarium Chemistry and the effect of Redox thereon much the same way many continued to claim the "earth was flat" even when proven incorrect.
I CHALLENGE these few aquarium keepers with little practical aquarium experience who still challenge this subject (and likely still think the world is flat) to then answer as to why my many controlled experiments where everything was equal, but for methods of controlling oxidative stress such as the use on constant mineral replenishers & UVC, resulted in longer life spans of fish and higher disease resistance!!! The facts are beyond the many citations here, there is much more information that is easily found as per the effects of oxidative stress in medical studies.
It is important to note, that just because many aquatic articles report only the importance of an oxidizing Redox, this does not make it 100% correct, science is not a vote! It really is not that difficult to read outside (non-aquarium hobby) articles that show otherwise. This is an unfortunate side of the aquarium hobby is that many will pass around old tired so-called facts as truths without reading anything that contradicts their theories masquerading as fact.
Often these same persons will use "Red Herring" deflection arguments since they cannot refute the information provided here and in the cited references. A common "Red Herring" argument is so-called peer reviews; unfortunately, when peers in this hobby refuse to understand the Redox Balance, despite growing information outside the hobby, how can there be peer review? HOWEVER, this article in particular has received MANY positive reviews/compliments from outside the industry such as Dr. Mamoon Kundi and his research of how Redox affects human health (and others cited within the article). As well I have been consulted by many within the industry/hobby including Disney for help with their public aquariums.
I read a recent article that is easily found in search that is admittedly well written dealing with Aquarium Redox, with a good explanation as to what Redox is and more, however this article still chooses to repeat much of the same tired old information about only the positives of the oxidative side of the Redox equation, missing the evidence that the reduction side is also VERY important to a healthy Aquarium Redox Balance (I noted that the sources this particular article sited were all more than 15 years old which may explain much of the out of date information).
Ignoring both sides of this equation is akin to acknowledging the importance of oxidation in the breakdown of dead animal and plant material, yet to ignore the fact that if this oxidation process were to continue unabated without reduction, we and all other living organisms would be consumed immediately!!! This simple but important point must be understood.
Part of the problem is that many hold onto thinking like this: "Poor water quality is equated with Low Redox Potential, whereas High Redox Potential is a measure of high-water quality and stability" when in fact as this article has shown, it is much more complex than this as we seek a healthy Redox BALANCE.
While admittedly a complex subject, the basics of Redox are really not that hard to understand and are easily applied; that is knowing that a high bio load can have a downward effect on a good Redox while water changes, additional mineralization, UV Sterilization and more can have positive effect on Redox.
Simply put, think of a Redox Balance of a magnet with opposite poles, and with without this electromagnetism of the water which makes up 90% of living tissue, the health of any living organism be it a fish, or aquatic plant will be affected!
You must realize that these electrons necessary for balancing Redox are often used up and the incorrect assumptions often made about GH, UV Sterilization simply do not fit the evidence! I will also note, the more complex aspect/equations of Redox are certainly worth learning (I am still trying to “grasp” them all), however I would not let it frustrate you if they are difficult to understand.
More simply stated; you can have too much oxidation, as well you can have too much reduction. However, based on my years of experience and feedback, most understand the undesirability of too much reduction (such as "too high" of a GH) but fail to understand the issues of too much oxidation which hopefully readers will now understand better.
Further Reading:
• Aquarium Chemistry; GH
Many aquarium keepers, especially advanced, are already applying many of the "best methods" for healthy aquarium redox without even knowing it. An example is an advanced aquascaper using the EI method: Who by water changes, adding GH booster, and avoiding the oxidative side of of the bio cycle by virtue of the plants performing this action, is already likely taking care of 80-90% of oxidative stress issues. However often even here it can be improved slightly with the addition of a true UV Sterilizer, constant GH maintenance methods, and improved moderately by switching to a fish food that optimizes energy levels!
Finally, maintaining a Redox below +350 mV while applying methods that might both permanently and temporarily achieve a Redox (low numbers are temporary) of about +350 to +100 mV for marine or +300 to -200 mV for freshwater are the raw numbers we are aiming for. (As per the graph in the background section).
However, if you have read this article thoroughly including the outside links/website other than my material, you will know that this is an extreme oversimplification as there are times for higher and lower Redox readings! Better would also be to go by rH numbers of about 23 to 28 (see the Relative Hydrogen Section of this article).
In other words, think of your Redox as constantly in flux with there certainly being times for a high/oxidizing Redox. In fact, if you were to measure your aquarium hourly with an ORP Meter (such as the AAP ORP/PH Meter) from lights on until an hour after lights off, you should observe different readings in most healthy aquariums. However, a +350 or higher Redox should NOT be the norm for most healthy aquariums or ponds.
Product Resource:
• AAP ORP METER
For further information about
• WHY YOU SHOULD USE A UV STERILIZER
Another good article about this subject that I found is this:
• ORP and the Reef Aquarium
Here is a quote from the above article: “Many aquarists have been led to believe that ORP is a measure of water quality or purity. Manufacturers selling ozonizers and other oxidizers (like permanganate) have been especially keen to present that idea. But is it really true? Is a higher Redox indicative of "purer water" even when that Redox is manipulated artificially by adding strong oxidizers? Or is such an addition analogous to an air freshener that masks odors? I don't know the answer, but I think that aquarists should ask the question, and hope to hear useful answers before adding such materials to their aquaria.”
This statement is where many aquarists have gone wrong in my opinion, A positive Redox is NOT a measure of water quality, but at least a Reducing Redox has more potential for a healthy aquarium.
More Resources/references (These are MUST reads in my opinion):
• Oxidative Stress Effects (May 2015)
• Nitroso–Redox Interactions in the Cardiovascular System
• The Relative Hydrogen Score, aka the rH Score (aka rH2 Score)
Abstract: Discusses a version of the Nernst equation posited by Clark in 1923 for computing a true relative hydrogen reducing power score (rH score, aka rH2 score) from examination of both pH and ORP.
• Redox Biology Center (RBC)
• Which Way Does Electricity Really Flow?
• Use of Higher Cation Fe+3 in Therapy
In fact, this is now probably my most researched subject, with many outside sources cited here. It is also noteworthy that many concepts applied here to explain my researched results are readily explained in what is now common knowledge as per oxidative stress in humans (this includes diet). As well, I have received emails from outside the aquarium community from medical researchers congratulating my conclusions as to Redox by citing current research and then applying it to aquariums.
Unfortunately, some aquarists in their writings or anecdotal forum comments fail to look at the research that has gone on the subject of Redox and stick to their old tired theories about Aquarium Chemistry and the effect of Redox thereon much the same way many continued to claim the "earth was flat" even when proven incorrect.
I CHALLENGE these few aquarium keepers with little practical aquarium experience who still challenge this subject (and likely still think the world is flat) to then answer as to why my many controlled experiments where everything was equal, but for methods of controlling oxidative stress such as the use on constant mineral replenishers & UVC, resulted in longer life spans of fish and higher disease resistance!!! The facts are beyond the many citations here, there is much more information that is easily found as per the effects of oxidative stress in medical studies.
It is important to note, that just because many aquatic articles report only the importance of an oxidizing Redox, this does not make it 100% correct, science is not a vote! It really is not that difficult to read outside (non-aquarium hobby) articles that show otherwise. This is an unfortunate side of the aquarium hobby is that many will pass around old tired so-called facts as truths without reading anything that contradicts their theories masquerading as fact.
Often these same persons will use "Red Herring" deflection arguments since they cannot refute the information provided here and in the cited references. A common "Red Herring" argument is so-called peer reviews; unfortunately, when peers in this hobby refuse to understand the Redox Balance, despite growing information outside the hobby, how can there be peer review? HOWEVER, this article in particular has received MANY positive reviews/compliments from outside the industry such as Dr. Mamoon Kundi and his research of how Redox affects human health (and others cited within the article). As well I have been consulted by many within the industry/hobby including Disney for help with their public aquariums.
I read a recent article that is easily found in search that is admittedly well written dealing with Aquarium Redox, with a good explanation as to what Redox is and more, however this article still chooses to repeat much of the same tired old information about only the positives of the oxidative side of the Redox equation, missing the evidence that the reduction side is also VERY important to a healthy Aquarium Redox Balance (I noted that the sources this particular article sited were all more than 15 years old which may explain much of the out of date information).
Ignoring both sides of this equation is akin to acknowledging the importance of oxidation in the breakdown of dead animal and plant material, yet to ignore the fact that if this oxidation process were to continue unabated without reduction, we and all other living organisms would be consumed immediately!!! This simple but important point must be understood.
Part of the problem is that many hold onto thinking like this: "Poor water quality is equated with Low Redox Potential, whereas High Redox Potential is a measure of high-water quality and stability" when in fact as this article has shown, it is much more complex than this as we seek a healthy Redox BALANCE.
While admittedly a complex subject, the basics of Redox are really not that hard to understand and are easily applied; that is knowing that a high bio load can have a downward effect on a good Redox while water changes, additional mineralization, UV Sterilization and more can have positive effect on Redox.
Simply put, think of a Redox Balance of a magnet with opposite poles, and with without this electromagnetism of the water which makes up 90% of living tissue, the health of any living organism be it a fish, or aquatic plant will be affected!
You must realize that these electrons necessary for balancing Redox are often used up and the incorrect assumptions often made about GH, UV Sterilization simply do not fit the evidence! I will also note, the more complex aspect/equations of Redox are certainly worth learning (I am still trying to “grasp” them all), however I would not let it frustrate you if they are difficult to understand.
More simply stated; you can have too much oxidation, as well you can have too much reduction. However, based on my years of experience and feedback, most understand the undesirability of too much reduction (such as "too high" of a GH) but fail to understand the issues of too much oxidation which hopefully readers will now understand better.
Further Reading:
• Aquarium Chemistry; GH
Many aquarium keepers, especially advanced, are already applying many of the "best methods" for healthy aquarium redox without even knowing it. An example is an advanced aquascaper using the EI method: Who by water changes, adding GH booster, and avoiding the oxidative side of of the bio cycle by virtue of the plants performing this action, is already likely taking care of 80-90% of oxidative stress issues. However often even here it can be improved slightly with the addition of a true UV Sterilizer, constant GH maintenance methods, and improved moderately by switching to a fish food that optimizes energy levels!
Finally, maintaining a Redox below +350 mV while applying methods that might both permanently and temporarily achieve a Redox (low numbers are temporary) of about +350 to +100 mV for marine or +300 to -200 mV for freshwater are the raw numbers we are aiming for. (As per the graph in the background section).
However, if you have read this article thoroughly including the outside links/website other than my material, you will know that this is an extreme oversimplification as there are times for higher and lower Redox readings! Better would also be to go by rH numbers of about 23 to 28 (see the Relative Hydrogen Section of this article).
In other words, think of your Redox as constantly in flux with there certainly being times for a high/oxidizing Redox. In fact, if you were to measure your aquarium hourly with an ORP Meter (such as the AAP ORP/PH Meter) from lights on until an hour after lights off, you should observe different readings in most healthy aquariums. However, a +350 or higher Redox should NOT be the norm for most healthy aquariums or ponds.
Product Resource:
• AAP ORP METER
For further information about
• WHY YOU SHOULD USE A UV STERILIZER
Another good article about this subject that I found is this:
• ORP and the Reef Aquarium
Here is a quote from the above article: “Many aquarists have been led to believe that ORP is a measure of water quality or purity. Manufacturers selling ozonizers and other oxidizers (like permanganate) have been especially keen to present that idea. But is it really true? Is a higher Redox indicative of "purer water" even when that Redox is manipulated artificially by adding strong oxidizers? Or is such an addition analogous to an air freshener that masks odors? I don't know the answer, but I think that aquarists should ask the question, and hope to hear useful answers before adding such materials to their aquaria.”
This statement is where many aquarists have gone wrong in my opinion, A positive Redox is NOT a measure of water quality, but at least a Reducing Redox has more potential for a healthy aquarium.
More Resources/references (These are MUST reads in my opinion):
• Oxidative Stress Effects (May 2015)
• Nitroso–Redox Interactions in the Cardiovascular System
• The Relative Hydrogen Score, aka the rH Score (aka rH2 Score)
Abstract: Discusses a version of the Nernst equation posited by Clark in 1923 for computing a true relative hydrogen reducing power score (rH score, aka rH2 score) from examination of both pH and ORP.
• Redox Biology Center (RBC)
• Which Way Does Electricity Really Flow?
• Use of Higher Cation Fe+3 in Therapy
Videos
|
|
|